Chronic Obstructive Pulmonary Disease (COPD)
Patient Management
Online Continuing Education Course

Course Description
COPD course on patient management. Continuing education course covering causes and forms of chronic obstructive pulmonary disease, characteristics, effects of smoking, breathing difficulty, emergency evaluation, long-term treatment, and management. Includes pulmonary rehabilitation and treatment of acute exacerbations. 6-contact-hour CEU for nursing and other healthcare professions.
Course Price: $39.00
Contact Hours: 6
Pharmacotherapeutic Hours: 0.5
Course updated on
May 2, 2025
"Excellent and informative. Clearly differentiates signs and symptoms." - Denise, OT in New York
"The course increased my knowledge of COPD and emphysema. It gave me clearer guidelines as to how to tackle some of the questions that arise in providing care to our patients." - Julie, RN in Guam
"I enjoyed this course, especially taking it in my own home. I get anxious when having to take a test, and so I was more relaxed at home to concentrate." - Betsy, OTA in Ohio
"Fantastic class. Excellent content, very well organized. Details were pertinent and easily understood." - Hannah, OT in Oregon
Chronic Obstructive Pulmonary Disease (COPD)
Patient Management
Copyright © 2025 Wild Iris Medical Education, Inc. All Rights Reserved.
LEARNING OUTCOME AND OBJECTIVES: Upon completion of this course, you will have increased your understanding of the causes of and the current treatments for chronic obstructive pulmonary disease (COPD). Specific learning objectives to address potential knowledge gaps include:
- Discuss the characteristics of COPD.
- Describe the physiology of normal lungs vs. lungs damaged by COPD.
- Identify the causes, functional effects, and preventive measures for COPD.
- Summarize the characteristic clinical findings in a patient presenting with COPD.
- Outline the lifestyle changes and pulmonary rehabilitation components of a long-term treatment plan.
- Describe pharmacologic, oxygen therapy, and surgical interventions for COPD.
- Discuss acute exacerbations of COPD and their treatment.
TABLE OF CONTENTS
WHAT IS COPD?
Chronic obstructive pulmonary disease is a preventable condition that makes it difficult to move air into and out of a person’s lungs. Difficulty moving air in the lungs is called airflow obstruction or airflow resistance. COPD is characterized by a progressively increasing airflow obstruction that cannot be fully reversed, although it can sometimes be temporarily improved by medications. In almost all cases, COPD has been caused by the long-term inhalation of pollutants, especially cigarette smoke (GOLD, 2025; NIH, 2024a).
COPD and other chronic lower respiratory diseases were the sixth leading cause of death in the United States in 2022. This is down from previous years, mainly because of a decrease in smoking, decreased mortality from ischemic heart disease and infectious disease, and advanced aging (CDC, 2024e). The tabulated cost of COPD was more than $31 billion on patient-care costs in the United States in 2019. These costs are projected to reach approximately $60 billion in 2029 (NIH, 2024b).
COPD develops quietly. Early in their disease, patients have measurable declines in their lung function before they develop symptoms. The first symptoms are usually an intermittent cough and some shortness of breath during exercise. Patients often dismiss these as temporary lung irritations or as a lack of physical conditioning. After many years, the cough becomes chronic or the spells of breathlessness become more frequent. Typically, this is the stage at which people first seek medical help.
The specific form that COPD takes falls along a spectrum. At one end of the spectrum, people get emphysema, and at the other end of the spectrum, people get chronic bronchitis. Many people with COPD have a mix of both emphysema and chronic bronchitis. (These two forms of COPD are discussed in detail below.)
Regardless of its form, COPD causes dyspnea (difficulty breathing). Dyspnea feels like shortness of breath. Initially, shortness of breath occurs only during vigorous exercise. Subsequently, the dyspnea begins to happen with mild exercise. Eventually, normal activities of daily living cause dyspnea. Finally, a person with COPD is short of breath even when at rest. This relentless increase of dyspnea gradually limits a person’s activities, and at some point, it becomes hard for a person with COPD to do anything but sit or lie down (Harding et al., 2022).
Patients with COPD have little to no reserve capacity or volume in their lungs, placing them at greater risk of developing hypoxemia. Hypoxemia occurs when peripheral oxyhemoglobin saturation (SpO2) (normal range 94%–100%) and arterial oxygen tension (PaO2) (normal range 80%–100%) are less than normal. This causes a reduction of oxygen in the blood.
Patients with COPD also exhibit hypercapnia, with a partial carbon dioxide (PaCO2) level greater than 45%. In earlier stages of COPD, hypercapnia may contribute to the person’s respiratory drive, helping to increase the depth and quality of breathing (Harding et al., 2022). If the PaCO2 rises too high, the brain is depressed, causing inadequate ventilation that can lead to a coma (Pagana et al., 2022).
Respiratory infections, increases in inhaled pollutants, and the occurrence of other medical diagnoses will further reduce the lungs’ ability to absorb oxygen and to expel carbon dioxide. These problems can send patients with COPD into hypoxemia. Such stresses are unavoidable, so patients with COPD suffer repeated episodes of significantly worsened symptoms, called acute exacerbations. Acute exacerbations are more frequent in patients who are susceptible to infections (GOLD, 2025). Acute exacerbations resolve slowly over weeks or months, even with medical treatment, and sometimes must be managed in a hospital.
After COPD has become symptomatic, the disease is treated with bronchodilators, which can ease the patient’s dyspnea so that a wider range of activities remains tolerable. Using the combination of a corticosteroid inhaler and a muscarinic antagonist can reduce the severity and duration of exacerbations and the occurrence of hospital admissions.
However, COPD follows a relentless downward course. Even with bronchodilator therapy and other treatment parameters, the patient’s lung function continues to gradually decline. Eventually, dyspnea limits a COPD patient to only minimal activity. Patients are continually fatigued, they lose weight, and at some point, they typically succumb to a respiratory illness, cor pulmonale, heart failure, renal failure, diabetes with neuropathy, or acute respiratory failure. Supplemental oxygen therapy can prolong some patients’ lives, and select patients can benefit temporarily from lung surgery (discussed later in this course).
Acute exacerbations continue for all patients, and most patients eventually succumb to an acute exacerbation that cannot be reversed (Harding et al., 2022).
ANSWERING PATIENT QUESTIONS
Q: What is COPD?
A: COPD is an abbreviation for chronic obstructive pulmonary disease. This disease is caused by inflammation of the lungs due to many years of breathing in cigarette smoke or other types of pollution. The airways in the lungs become narrowed, and in some people, the airways become clogged with mucus. These problems make it harder and harder to move air into and out of the lungs.
A person with COPD frequently feels short of breath. COPD makes normal breathing tiring, and it can make it so difficult to breathe that exercise becomes too tiring to do. COPD continues to worsen over time, especially if the person is still smoking.
Q: How do I know if I have COPD?
A: The signs and symptoms of COPD are different for each person, but common symptoms are cough, coughing up mucus, shortness of breath, wheezing, and chest tightness. COPD usually occurs in people who are at least 40 years old and who have smoked for many years. To make the diagnosis, a provider will administer a physical exam and a set of breathing tests.
Q: Once I have COPD, what can I do to fix it?
A: COPD cannot be cured, but it can be treated to make your life more comfortable. See your primary care provider and get set up with a treatment plan tailored specifically for you. Meanwhile, quitting smoking is the single most important thing you can do to slow the progress of the disease.
Airflow Obstruction: The Essence of COPD
In the past, patients with COPD with emphysema were said to have type A COPD and were sometimes called pink puffers (normal PaCO2, PaO2 >60 mmHg, emphysematous lung tissue destruction). Patients with COPD with chronic bronchitis were said to have type B COPD and were sometimes called blue bloaters (PaCO2 >45 mmHg, PaO2 <60 mmHg, pulmonary hypoxic vasoconstriction) (McMillen, 2023a).
These names are no longer used because the division of COPD into two alternative types is too simple. Many patients have a mix of emphysema and chronic bronchitis. Currently, the emphasis is on the common feature of all patients with COPD: airflow obstruction. Whether it appears as emphysema, chronic bronchitis, or a mixture of the two, COPD is characterized by chronic, worsening, and irreversible airflow obstruction (Harding et al., 2022).
Prevention
COPD can be almost entirely prevented by avoiding long-term inhalation of pollutants, mainly cigarette smoke. As they age, all people suffer a decline in lung function. Smokers who quit before developing symptoms of COPD can often reduce the decline in lung function to nearly normal levels within a few years of remaining smoke free, although established damage will not improve (GOLD, 2025).
COPD INCIDENCE
COPD is the most common serious lung disease in the United States. Over the last few decades, there has been an increase in the percentage of Americans with COPD. Currently, 17 million adults (4.6%) in the United States have a diagnosis of COPD. An unknown number of Americans with COPD may still be undiagnosed. Among people with COPD, most people have a combination of the chronic bronchitis form and the emphysematous form (ALA, 2024a; NIH, 2024b).
Age of Onset
Seventy-five percent of deaths of patients with COPD are secondary to excessive smoking, although 25% of those with COPD have never smoked (NIH, 2022). The characteristics of the population of people with COPD—such as chronic cough, shortness of breath, limited energy, and sputum production—are the same as the characteristics of the population of people who have been longtime smokers.
COPD is most common in older people because symptomatic COPD usually takes more than 20 pack-years (see below) of smoking to develop. In 2022, a lower percentage of adults ages 40–64 years (5.4%) had any lung obstruction compared with adults ages 65 years or older (9.7%). Approximately 15% of adult Americans are smokers (ALA, 2024a).
The current generation of older adults has done a record-breaking amount of cigarette smoking. Although many older Americans have stopped smoking, even those who quit can develop symptoms of COPD and suffer a greater-than-normal decline in their breathing ability late in life. Physiologic changes in older adults with COPD, including reduced lean muscle mass and decreased respiratory muscle strength, cause an increased disease burden, including more frequent exacerbations, more dyspnea, and lower exercise tolerance. The higher incidence of comorbidities, especially cardiac, may prove a more challenging plan of treatment, for instance, due to potential drug-drug interactions. Impaired cognition, arthritis, impaired vision and hearing, and depression will also affect disease progression and prognosis (Harding et al., 2022).
Almost 90% of adults who smoke cigarettes daily first tried smoking by age 18. Tobacco products are now flavored to appeal to youthful smokers. In 2024, 88.2% of high school students and 85.7% of middle school students who used tobacco products reported using a flavored e-cigarette. At the current rate of smoking in those under age 18, 8.6 million will die from smoking-related diseases (CDC, 2024a).
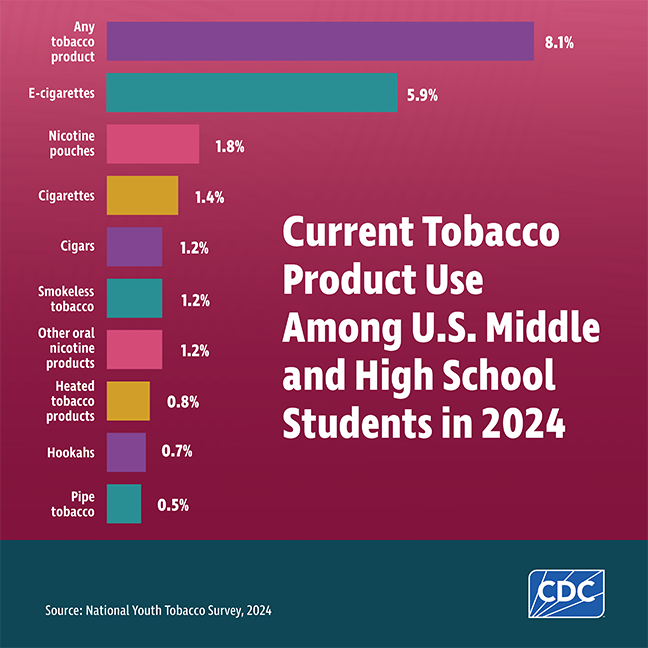
(Source: National Youth Tobacco Survey, 2024; CDC, 2024a.)
(See below for more information about e-cigarettes, or “vaping.”)
PACK-YEARS
A person’s smoking intensity is measured in pack-years. The typical patient with COPD has a smoking history of more than 40 pack-years. One “pack-year” means that a person has smoked approximately one pack (20 cigarettes) per day for one year. Smoking one half pack a day for one year is equivalent to one half pack-year, and smoking two packs a day for 10 years is equivalent to 20 pack-years.
Sex
Women are diagnosed with COPD later than men, causing the disease to be more advanced and therefore any treatment to be less effective. Women have an increased susceptibility to COPD caused by smoking, likely due to smaller lungs, smaller airways, and lower elastic recoil. Women who smoke are 50% more likely to develop emphysema. Women with COPD experience a worse quality of life, more acute exacerbations, more dyspnea, and less secretions, but a better response to oxygen therapy than men. Women comprise 56% of the people with COPD and men 44% (Harding et al., 2022; NIH, 2024b; Tomashaw & Carter, 2023).
Race
The prevalence of COPD follows the history of the level of smoking in a population. In the United States, higher rates of COPD are found among those who have had the highest levels of smoking. The majority of all people with COPD are current or former smokers. By race/ethnicity, the prevalence of COPD among American Indians/Alaskan Natives is 9%, African Americans 5%, Whites 5%, Hispanics 2%, and Asians 1% (NIH, 2022).
Mortality Rates
The increased level of smoking by women over the past 30 years is causing women’s death rates from COPD to rise, while the death rates from COPD is dropping for men. The greater mortality in women may also be due to their smaller lungs, the contribution of estrogen to the severity of lung disease in women, and misdiagnosis in women. COPD and other chronic lower respiratory diseases are the sixth-leading cause of death in the United States (CDC, 2024e).
PATHOPHYSIOLOGY OF COPD
In COPD, the body’s reaction to inhaled pollutants (mainly smoke) results in chronic inflammation of the bronchial tree. Inflammation is a natural protective reaction, but it is useless against air pollutants. Instead of helping, the persistent inflammatory reactions damage the lungs.
Normal Lungs
Understanding the structure and function of normal lungs is helpful to understanding COPD’s inflammatory damage.
LUNG STRUCTURE
The two lungs comprise approximately 3 million microscopic alveoli clustered at the ends of tiny air tubes. The lung tubes begin at the trachea and branch into successively narrower, shorter, and more numerous tubules. The central tubes are the bronchi and bronchioles. The most peripheral tubes are the respiratory bronchioles, which are lined with alveoli. It is through the walls of the alveoli that gases are exchanged between the inspired air and the blood in the surrounding capillaries.
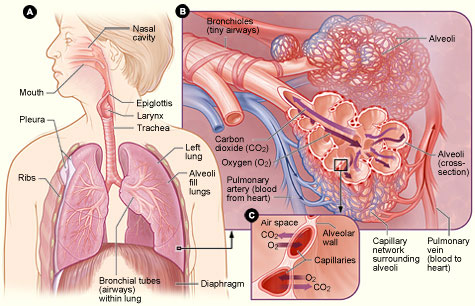
Figure A: Locations of the respiratory structures in the body.
Figure B: Enlarged image of airways, alveoli, and their capillaries.
Figure C: Location of gas exchange between the capillaries and alveoli.
(Source: National Institutes of Health.)
The medium and large bronchi are wrapped with smooth muscle, which tightens to narrow the airways and relaxes to widen the airways. The walls of all the airways are lined by ciliated epithelial cells with interspersed secretory cells, which coat the inner walls of the airways with mucus. All of the cilia of the pseudostratified epithelial cells beat in the direction of the trachea and throat, so mucus and trapped particles are continuously moved up and out of the lungs.
Healthy lungs are lightweight, soft, spongy, and elastic. The anatomy of the lungs promotes gas exchange between the circulatory system and the source of oxygen in air. Normally, the chest walls stretch the lungs and keep them expanded to three times their relaxed size. When the chest is surgically opened, however, the lungs recoil as the innate elasticity of the lungs pulls them back to their resting size (Haddad & Sharma, 2023).
When an adult takes a full breath, the volume of air in the lungs is about 6 liters on inspiration. During life, the lungs are never completely empty; even after a complete exhalation, there are about 2.5 liters of air left.
LUNG FUNCTION
Lungs are the organs through which oxygen is absorbed into and carbon dioxide is expelled from the bloodstream. These gas exchanges occur through the walls of the alveoli and the terminal respiratory airways, which make up the distal-most air spaces inside the lungs.
Maintaining healthy levels of blood gases is the lungs’ primary function, and the lungs contain an extensive capillary system to provide more than the necessary surface for gas exchange. The lung tissue itself is very thin and delicate, and most of the volume inside a normal lung is taken up by air. Since lung tissue is thin and air is light, most of the weight of a lung can be attributed to the blood circulating in it (Haddad & Sharma, 2023).
People with healthy lungs rarely use all the gas-exchange potential of their lungs. During the most strenuous activity, a healthy person will use only 60%–70% of their maximal ventilatory capacity. Strenuous exercise does cause temporary dyspnea, but the 30%–40% ventilatory reserve quickly relieves the dyspnea of a healthy person after a short rest. Even the dyspnea caused by strenuous exercise in a healthy person is not as debilitating as the dyspnea in a person with severe COPD.
Healthy lungs function less efficiently as they age. As people get older, their chest walls stiffen, bones become weaker, and respiratory muscles weaken. These changes make breathing almost twice as much work for a 70-year-old as for a 20-year-old. The forced vital capacity (VC or FVC) and the amount of air that can be exhaled in one second (1-second forced expiratory volume, or FEV1) gradually and progressively decline during a person’s lifetime. In a healthy person, none of these natural lung changes approaches the dramatic declines caused by COPD. The natural decline in lung function worsens the already-compromised breathing of those older adults who have COPD (Haddad & Sharma, 2023; Harding et al., 2022).
Lungs with COPD
COPD slowly destroys the lungs and makes it increasingly difficult for a patient to breathe. The most serious effect of COPD is a progressive obstruction of airflow. In COPD the airways leading into the alveoli become narrowed and less flexible, and they are often clogged with mucus. Eventually, many alveoli coalesce into larger, useless air spaces because the walls separating the alveoli become damaged or destroyed.
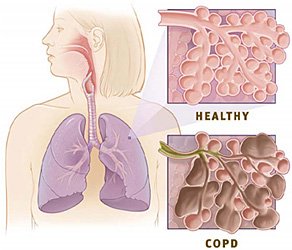
Upper right: Healthy alveoli.
Lower right: Alveoli with COPD.
(Source: National Institutes of Health.)
The specific form that COPD takes varies from person to person. The two predominant forms of COPD are emphysema (destruction of alveoli) and chronic bronchitis (inflammation of the conducting air tubules).
EMPHYSEMA
For some people, COPD causes significant destruction of the terminal airways and air alveoli. This form of COPD is called emphysema. In emphysema, the overall architecture of the lung is altered dramatically, and the lung becomes honeycombed with useless spaces. These air spaces are created when the walls of the small respiratory airways and their alveoli are torn, allowing neighboring airways and alveoli to merge.
In the process, the surrounding capillaries become damaged, resulting in reduced pulmonary perfusion. Another characteristic of emphysema is decreased elasticity of lung tissue, resulting in reduced recoil and increased stiffness. Besides reducing the lung area available for gas exchange, emphysema leads to hyperinflated lungs and obstructed airflow (Harding et al., 2022).
CHRONIC BRONCHITIS
The other main type of COPD involves inflamed airways that become clogged with mucus produced by the goblet cells in the lungs. Patients with this variant of COPD develop a chronic cough that brings up sputum. This manifestation of COPD is a form of chronic bronchitis, which is defined as a persistent mucus-filled cough that has occurred frequently for at least three months per year in two consecutive years and that is not caused by another disease such as an infection, cancer, or congestive heart failure. It is characterized by an increase in the number and the size of mucous glands in the airways of the lung.
Chronic bronchitis can occur without COPD. More than one third of smokers have chronic bronchitis, but the disorder is only considered a form of COPD when there is also significant obstruction to airflow within the lungs (Harding et al., 2022).
Contributors to COPD
Cigarette smoking is considered one of the biggest health problems facing the world. In the industrialized world, cigarette smoking is the main cause of COPD. In underdeveloped countries, smoke from plant products that are burned for indoor cooking or heating is as much a cause of COPD as is cigarette smoking.
In 2020, the global prevalence of smoking was 22.3% of the total population, separated into 36.7% of men and 7.8% of women. This equates to approximately 1.3 billion people. There are more than 9 million deaths annually due to smoking—approximately 8 million as a direct result of smoking cigarettes and 1.3 million a result of secondhand smoke. Fifty percent of those who don’t quit smoking will die as a result of smoking. In the United States the death toll due to smoking is estimated to be 480,000 every year (WHO, 2023).
In the United States, chronic lung diseases, including COPD, account for 75% of smoking-related conditions. People who have never smoked account for 25% of all COPD cases. Other smoking-related diseases or conditions include throat cancer, stroke, heart attack, and asthma. Other irritants that may cause COPD are air pollution, environmental or workplace chemical fumes or dusts, and secondhand smoke (NIH, 2022; Su et al., 2024).
Many long-term smokers eventually develop COPD, but the severity of the disease varies from person to person, even among heavy smokers. People living in the same environment and smoking the same amount can differ in their propensity for developing COPD. Two factors have been suggested as the basis for this difference: individual physical characteristics and genetic factors (Harding et al., 2022). The longer and more intensely people smoke, the more likely they are to develop COPD.
The vapor from electronic cigarettes (e-cigarettes, also referred to as e-cigs, e-hookahs, mods, vape pens, vapes, tank systems, and electronic nicotine delivery systems [ENDS]) also contains substances that are known to be harmful. These include heavy metals (tin, nickel, and lead), nicotine, carcinogenic chemicals, ultrafine particles causing tissue irritation, and volatile organic compounds (CDC, 2024b).
The possible negative effects of e-cigarettes depend on the components contained within the device, including vitamin E acetate in some e-cigarettes containing tetrahydrocannabinol (THC). A CDC study of e-cigarette or vaping product use–associated lung injury (EVALI) showed an increase in inflammation in the lungs and impaired gas exchange caused by the aerosolized oils within the e-cigarettes. Most of the e-cigarettes that cause these types of injuries involve THC as the active ingredient (ALA, 2024b).
The effects of vaping by persons already diagnosed with COPD include additional inflammation of lung tissue. The chemicals in e-cigarettes cause a toxic response in the lung tissue and reduce the ability of the lungs to respond defensively to viral infections. Many of the flavors contained in the aerosolized liquid in e-cigarettes produce substances that have deleterious effects on human embryonic stem cells and human pulmonary fibroblasts. A partial list of these chemical substances includes cinnamaldehyde, diacetyl, 2,3-pentanedione, acetoin, and benzoic acid (ALA, 2024c).
Multiple studies have been conducted regarding the effects on persons with COPD who smoke marijuana through the use of e-cigarettes. It was thought that there might be some benefit with additional bronchodilation for those with COPD from vaping marijuana. It is considered less harmful to the lungs than more traditional methods of smoking the drug but can still cause an injury. In fact, THC products have been found to play a role in lung injury caused by the use of e-cigarettes and regular cigarettes (ALA, 2024b).
People who experience extensive exposure to secondhand smoke (SHS) have a considerably higher occurrence of respiratory symptoms, asthma, and COPD. In a longitudinal study of 6,519 participants in Taiwan with no prior exposure to cigarette smoke, 7% of the group exposed to secondhand cigarette smoke for 48 months developed COPD compared to 4% of the nonexposure group. Similar studies in other countries, although with smaller study samples, show the same compelling results, with a direct correlation between extended exposure to SHS and the eventual development of COPD (Su et al., 2024).
INFLAMMATORY RESPONSE
Cigarette smoking causes COPD by inciting a chronic inflammatory response to the pollutants in the smoke. This persistent inflammation is caused by the release of proteases in the lungs and eventually leads to destruction of lung tissue, accumulation of mucus, and thickening of small airways. Smoke also flattens the cilia in the airways and prevents them from removing mucus and fluid. Prolonged pulmonary inflammation is eventually accompanied by systemic inflammation. Factors such as inhaled pollutants, sedentary lifestyle, and infections may also contribute to systemic inflammation in someone with COPD.
The severity of inflammation may necessitate the use of corticosteroids. Patients with COPD are more resistant to the effects of corticosteroids, requiring higher doses and more prolonged use than more healthy smokers or nonsmokers. When treatment of COPD with steroids requires prolonged usage or higher dosages, COPD patients are more susceptible to developing pneumonia. Some COPD patients are resistant to steroids, either requiring a higher dosage or becoming unable to take steroids effectively (Harding et al., 2022).
Destruction of Lung Tissue
Lungs with COPD produce less enzymes that promote the formation of myofibroblast cells to aid in the healing of wounds and tissue. In the absence of this enzyme (alpha–smooth muscle actin, or α-SMA), diseased lung tissue in COPD is repaired more slowly (GeneCard, 2024). The progressive destruction of lung tissue leads to the emphysematous form of COPD, which is characterized by:
- Destruction of alveoli
- Loss of lung elasticity
- Loss of lung supporting tissue
- The collapse of small airways
(Harding et al., 2022)
Thickening of Small Airways
The hallmark of COPD is the increased resistance it causes for airflow in the lungs. In the chronic bronchitis form of COPD, much of the airflow obstruction comes from a progressive thickening and stiffening of the small airways in addition to increased mucus production.
The pathologic process underlying the narrowing of airways is fibrosis. With fibrosis, excess collagen accumulates in and around the airways, making them thicker and more rigid. Extra collagen is secreted as a natural repair response to tissue damage. Actins such as α-SMA are proteins related to cell motility, structure, integrity, and intercellular signaling. In COPD, myofibroblasts produce lower-than-normal levels of α-SMA, inhibiting the natural repair response (GeneCard, 2024).
The chronic bronchitis form of COPD includes changes in the small airways. These changes reduce airway volume. Specifically:
- Mucous cells proliferate and become larger; this generates excess mucus.
- The smooth muscle in the airway walls thickens.
- The airway walls bulge with invading inflammatory cells.
AIRWAY SENSITIVITY
People differ in their airway sensitivities, that is, in how readily their airways constrict when exposed to a variety of irritants such as pollen, dust, and chemicals. Asthma is the most common disease of people who have abnormally sensitive airways. People with COPD also tend to have sensitive and reactive airways. Although asthma and COPD are different diseases, smokers with asthma or with the tendency to develop asthma are more likely to develop COPD and are more likely to have COPD that worsens quickly (Harding et al., 2022).
ALPHA-1 ANTITRYPSIN (AAT) DEFICIENCY
Besides airway sensitivity, certain genetic factors make some individuals especially susceptible to developing COPD. One of these genetic propensities is an autosomal recessive disorder called alpha-1 antitrypsin deficiency. The gene for AAT is recessive. Therefore, someone with one normal and one faulty allele for the deficiency would be a carrier but not more susceptible to COPD. People of northern European descent are affected the most.
AAT deficiency allows the chronic inflammation caused by inhaled smoke to do considerable damage to the lungs; specifically, AAT deficiency fosters the destruction that causes emphysema. AAT is produced by the liver, and a deficiency can produce liver as well as lung disease.
Longtime smokers typically develop COPD when they are 50 to 60 years old, but smokers who are born with AAT deficiency develop symptomatic COPD 10 to 20 years earlier, at an average age of 40 years. Emphysema can even develop in nonsmokers if they have a severe AAT deficiency (GOLD, 2025; Harding et al., 2022).
Treatment is the replacement of the deficient AAT with the drug Prolastin (alpha).
ANSWERING PATIENT QUESTIONS
Q: What causes COPD?
A: Smoking is the most common cause of COPD. Cigarette, cigar, and pipe tobacco can all cause COPD when the smoke is inhaled. Other kinds of air pollution can be just as bad as smoke if the pollution is inhaled for many years. Anyone can get COPD from smoking, although it usually takes many years of smoking for the disease to be noticeable. A small number of people also have an inherited genetic defect called AAT deficiency.
Q: Can COPD be inherited?
A: Most types of COPD are not inherited. COPD is usually caused by cigarette smoking. Teaching children not to smoke will help protect them from getting COPD. A small number of people inherit a genetic defect called AAT deficiency, which makes them more likely to get the disease after only a few years of smoking or sometimes without having ever smoked at all. When these people get COPD, it is the emphysema type of COPD, and it usually shows up early, in people younger than 40 years old.
Q: Is COPD contagious?
A: No.
Functional Effects of COPD
REDUCED FEV1
When inhaling, a person stretches their chest and lung tissues. During exhalation, the elastic recoil of the chest and lungs is a major contributor to the force that pushes air out of the lungs. Failure to force a sufficient amount of air out of the lungs upon expiration causes the buildup of carbon dioxide (CO2) in the lungs and then in the blood (hypercapnia).
In COPD, fibrosis reduces lung elasticity. Therefore, a patient with COPD needs to replace the lost elastic force with extra muscular effort, and the extra effort must be sustained for a longer time. The narrowed airways in lungs with COPD carry smaller volumes of air, and people with COPD take longer to empty their lungs.
The extent of airway obstruction can be quantified for patients with COPD. One standard assessment measures the patient’s 1-second forced expiratory volume (FEV1), the volume of air that can be pushed out of the lungs during the first second after a full inhalation after the patient has taken a bronchodilator. A persistent, irreversible low FEV1 (≤70%) is the most characteristic objective finding in COPD (GOLD, 2025; Harding et al., 2022). (See “Lung Function Tests” below.)
HYPERINFLATION OF THE LUNGS
In COPD, the difficulty of breathing is worsened by excessively expanded (hyperinflated) lungs. Most people with COPD have some degree of emphysema, and part of each breath flows into nonfunctioning spaces in the lungs, where the air is unusable. To get sufficient oxygen into their system, people with COPD need to take larger breaths.
People with COPD also take longer exhaling, and after taking a large breath, in spite of the use of accessory muscles of respiration, there is not enough time to fully exhale the air. Excess air remains in their lungs during each breathing cycle.
Wasted air space and excess residual air lead to hyperinflated lungs. Hyperinflated lungs change the shape of the chest and diaphragm (described as barrel chest), making the mechanics of breathing more difficult. With hyperinflated lungs, breathing can be exhausting (GOLD, 2025).
HYPOXEMIA AND HYPERCAPNIA
Together, the obstructed airflow and the hyperinflated lungs of COPD make breathing arduous work. When COPD is severe, just the breathing required for slow walking could use one third of the body’s total oxygen intake.
In COPD, patients may not have enough energy to pull in all the oxygen they need or to expel all the carbon dioxide they produce. Compounding the problem of maintaining adequate gas exchange, COPD destroys alveoli and the small capillaries that surround them, making each breath even less effective.
As a result, people with severe COPD become chronically hypoxemic (too little circulating oxygen in the blood) and hypercapnic (too much circulating carbon dioxide in the blood). People with moderate COPD become hypoxemic during modest exercise, and as the disease worsens, they can become unable to exercise at all (Harding et al., 2022).
DYSPNEA AND ITS SPIRALING EFFECTS
Over the years, patients with COPD become less and less able to perform even modest exercise without developing dyspnea. Dyspnea is the most frequently reported symptom in patients with moderate and severe COPD, reported by >40% of such patients. The degree of dyspnea, as measured by the modified measured Medical Research Council (mMRC) Breathlessness Scale, is self-reported by the patient, similar to pain levels (see table below).
| Grade | Patient report of symptom severity |
|---|---|
| (UKRI, 2022; used with permission of the Medical Research Council.) | |
| 0 | I only get breathless with strenuous exercise. |
| 1 | I get short of breath when hurrying on level ground or walking up a slight hill. |
| 2 | On level ground, I walk slower than people of my age because of breathlessness, or I have to stop for breath when walking at my own pace on the level. |
| 3 | I stop for breath after walking about 100 yards or after a few minutes on level ground. |
| 4 | I am too breathless to leave the house, or I am breathless when dressing/undressing. |
The symptom burden for patients with COPD can be compared to the symptom burden of lung cancer patients. It comes from a mix of three sensations:
- The urge to breathe. This sensation is triggered by exercise or by the metabolic results of exercise: hypoxemia, hypercapnia, and metabolic acidosis.
- Difficulty breathing. This sensation is produced by excess chest movement and by unusual effort required by the muscles of respiration during breathing.
- Anxiety. This sensation can be caused by a fear of suffocating or by a memory of past discomfort with breathlessness. The anxiety of dyspnea can also come from entirely dissimilar sources of stress that are happening at the time, such as emotional and psychosocial factors like anxiety or depression.
(GOLD, 2025)
Breathlessness is upsetting. It stops people from exercising, and it is the main reason that people with COPD limit their activities. Dyspnea with exercise gets worse as COPD progresses. The degree of perceived breathlessness is proportional to respiratory effort. Exertional dyspnea may be caused by hyperinflation of the lungs from trapped air, resulting in reduced inspiratory volume. Patients begin to spend all their time either sitting in a chair or lying in bed, and after months of inactivity, patients with COPD become deconditioned as their muscles and circulatory system settle into sedentary states.
The pain and dyspnea that accompany COPD make it even more difficult to exercise. It is a spiraling problem: dyspnea causes lack of exercise, lack of exercise causes deconditioning, and deconditioning makes it harder to exercise. When they have become deconditioned, patients with COPD experience severe leg tiredness and leg pain when they try to exercise. Leg problems become yet another limiting factor when deconditioned people with COPD attempt to exercise (GOLD, 2025).
To break this cycle, people with COPD must exercise. Pulmonary rehabilitation, which includes gradually increasing, supervised training regimens, can reverse muscle weakness, reduce leg pain, and increase exercise tolerance (ALA, 2023b). (See “Pulmonary Rehabilitation” later in this course.)
Damage Beyond the Lungs
Patients with COPD have problems with organ systems other than their lungs. COPD leads to chronic hypoxemia, drains energy reserves, and is a source of chronic inflammation. These problems cause total-body muscle weakness and weight loss.
Chronic hypoxemia strains the heart and reduces the ability of the heart’s ventricles to respond to the demands of exercise. This may lead to ischemic tissue and heart failure. Caution must be taken in the pulmonary rehabilitation phase of treatment to prevent symptomatic ischemia.
Chronic inflammation initiates a generalized prothrombotic condition in the circulation. This makes blood clots more likely to form, and patients with COPD are at increased risk for developing myocardial infarctions (MIs), cerebral vascular accidents (CVAs), deep vein thromboses (DVTs), and pulmonary emboli (PEs).
CASE
Marilyn, a 72-year-old woman, presents to the emergency department with shortness of breath; tachypnea; and pain, heat, and redness in her right calf. She has a 40-pack-year history of smoking and quit 10 years ago when she was diagnosed with COPD. Contrast venography is performed to the right leg, and the radiologist diagnoses deep vein thrombosis. During the history taking, the patient states she just returned from a trip to visit her grandchildren, who recently moved across the country, and that the leg pain started soon after the long airplane flight.
The emergency department nurse explains to Marilyn and her family that patients with COPD are at higher risk for DVTs due to the chronic inflammation in the blood vessels caused by cigarette smoking. Once the COPD process starts, quitting smoking does not improve the problem.
Marilyn will be admitted to the hospital and started on anticoagulant therapy to prevent more clots from forming and on an exercise regime initiated by physical therapy.
PULMONARY HYPERTENSION IN COPD
Changes caused by COPD increase the arterial resistance inside the lungs. These include:
- Destruction of lung capillaries
- Thickened walls of small pulmonary blood vessels
- Constricted lung arteries due to chronic hypoxia and acidemia (a blood pH of <7.35 caused by greater-than-normal concentration of hydrogen ions)
- Constricted lung arteries due to the physical pressure of hyperinflated lungs
Due to these changes, more force is needed to push blood through the lungs, and the person develops pulmonary hypertension. Pulmonary arterial hypertension (PAH) is chronic, is progressive, and results in an increased pulmonary arterial pressure because of the constriction on the pulmonary blood vessels caused by alveolar hypoxia. In a normal adult lung, the mean pulmonary artery pressure is 12–16 mmHg. In a lung with pulmonary hypertension, the mean pulmonary artery pressure is >25 mmHg at rest and >30 mmHg with exercise.
Pulmonary hypertension is especially hard on the right ventricle of the heart, which hypertrophies in response. Pulmonary hypertension can exist comorbidly with other diseases such as heart failure and COPD. As the strain on the right ventricle persists, the heart can fail. Heart failure secondary to lung problems is called cor pulmonale, and COPD is the leading cause of cor pulmonale as a late manifestation of the lung disease (Harding et al., 2022). The incidence of mild pulmonary hypertension in COPD is 30.2% and of severe pulmonary hypertension in COPD is 7.2% (Zhang et al., 2022).
DEPRESSION
People with COPD have a high incidence of clinical depression and anxiety. The presence of these two psychological clinical characteristics increases mortality in COPD patients. The depression is not only a psychological reaction to their increasingly restricted lifestyles. There is a demonstrated increase in patients with COPD with depression when there is evidence of clinical determinants such as younger age, female sex, lower FEV1 (forced expiratory volume), higher St. George’s Respiratory Questionnaire (SGRQ) score (which measures the impact of obstructive airway disease on health and daily life), cough, and cardiovascular disease. COPD patients with depression may benefit from exercise, similar to other types of patients. COPD patients are two times more likely to die by suicide (GOLD, 2025).
CLINICAL APPEARANCE OF STABLE COPD
The Typical Patient with COPD
The “typical” American patient with moderate to severe COPD is a White female over 65 years of age with a history of smoking at least one pack of cigarettes a day for more than 40 years (i.e., 40 pack-years). She complains of general tiredness and becomes short of breath when exercising. Her legs bother her while walking, so she spends most of her time sitting. If asked to exhale quickly, it takes her an unnaturally long time.
Other aspects of the “typical” picture range along a spectrum:
- If this person is on the emphysematous end of the spectrum, she will tend to be thin and have a wide, barrel-shaped chest. She will always feel a great deal of dyspnea. When she coughs, she will not produce much sputum. On chest examination, this person’s breath sounds will be distant and relatively clear.
- If this person is on the chronic bronchitis end of the spectrum, she will tend to be of normal weight or overweight. She will cough frequently and will bring up sputum. On chest examination, her breath sounds will include rales (dry crackles), rhonchi (harsh, wet sounds), and wheezes. A COPD patient with chronic bronchitis has exacerbations usually related to bacterial respiratory infections.
(Harding et al., 2022)
Chief Complaints
Patients with COPD usually present with the complaints of dyspnea and coughing.
DYSPNEA
Dyspnea during mild exercise is the most common reason that people with COPD first seek out a physician. This dyspnea will have appeared gradually over a period of years. The dyspnea of COPD reflects at least two sensations:
- The urge to breathe. Patients with COPD have airway obstruction, and they cannot fully empty their lungs before they need to take another breath. The residual air, which keeps the lungs hyperinflated, dilutes the oxygen content of the newly inhaled air. Thus, these people feel hypoxemic.
- Difficulty breathing. Patients with COPD have hyperinflated lungs. Their chests remain overly expanded in the resting state (i.e., after exhaling). It is difficult for the respiratory muscles to expand their chest farther when attempting to take a new breath. Thus, these people put an unusual effort into breathing.
Sometimes a patient with COPD will come to the healthcare provider reporting that a recent illness has triggered dyspnea. Illnesses, especially respiratory illnesses and infections, worsen dyspnea. If the patient actually has COPD, a careful review of the history of the patient’s exercise tolerance usually turns up evidence of increasing dyspnea before the illness (Harding et al., 2022).
COUGH
While dyspnea is the symptom that most often brings patients with COPD to visit a healthcare provider, coughing is the most common symptom found in patients with early COPD. The patient who smokes may at first assume the early coughing is a result of the irritants in the tobacco. The cough of COPD is usually worse in the mornings. Early in the disease, the cough produces only a small amount of colorless sputum (i.e., mucus and lung secretions that are expelled into the throat by coughing), except in the case of chronic bronchitis.
Coughing typically begins earlier in the development of COPD than dyspnea, especially with chronic bronchitis, but unlike dyspnea, coughing may or may not limit the patient’s daily activities; it depends on what the patient needs to do in a day. For example, if they “teach or preach,” coughing may interfere with their work.
Coughing is stimulated by irritation of the bronchial tree. The sudden onset of new coughing is usually caused by irritation from a respiratory infection and is accompanied by fever, tachycardia, and tachypnea. This type of cough typically lasts less than three weeks, although in some people coughs can hang on as long as two months after a respiratory illness. The coughing of COPD, however, occurs intermittently for years (GOLD, 2025).
CASE
Shelley Bradley made an appointment with Maria, her family nurse practitioner (FNP), because of increased dyspnea after a viral respiratory infection she came down with in spite of getting her annual flu shot. She told Maria that she has had a persistent cough for three weeks after the first flu-like symptoms appeared.
Ms. Bradley was diagnosed with COPD four years ago. She quit smoking at that time and has a 32-pack-year history of smoking. She has no signs of infection and undergoes a chest X-ray, which shows no infection and no change in her airway. She is given a prescription for an ipratropium (Atrovent) inhaler to use in addition to her longer-acting salmeterol (Serevent) inhaler. She is instructed to exhale deeply before administering the medication and to hold her breath after each inhalation of the medication. Maria has her return the demonstration to show she understands proper technique.
Maria discusses with Ms. Bradley the importance of protecting herself from contracting a respiratory infection in the future. She recommends that Ms. Bradley wear a surgical mask when in a crowd, such as on an airplane or using public transit, to protect herself from contracting a respiratory infection, since COPD now makes her more vulnerable to respiratory infections and less able to recover. Maria discusses the availability of pneumonia vaccines and the value of frequent handwashing and avoiding proximity to people with signs of respiratory infections. She also discusses Ms. Bradley’s medication regimes before the end of the appointment.
Medical History
HISTORY OF THE CHIEF COMPLAINT
Almost as a rule, the health system first sees patients with COPD when they are in their late 40s to mid-50s and with chief complaints of dyspnea and excessive coughing. In retrospect, their symptoms have been going on for at least a decade, with coughing having shown up first. At one time, the dyspnea had been noticed only during heavy exertion, but eventually it began to interfere with even mild everyday activities. In the late stage of COPD, dyspnea may be continuous even when the person is at rest.
A thorough medical history of a COPD patient includes risk factors, previous medical history, pertinent family history, evidence of genetic factors, history of symptom progression, prior exacerbations and hospitalizations, current medications and allergies, comorbidities, and support available to the patient (CDC, 2024d; GOLD, 2025). During the medical history, most patients with COPD state that typical symptoms are exacerbated upon arising, usually in the morning. These symptoms may include, in descending order of occurrence:
- Dyspnea
- Inability to accomplish full expiration, resulting in air trapping
- Sputum
- Cough
- Wheezing
- Chest tightness
- General muscle weakness
The air trapping results in hyperexpansion of the chest (barrel chest).
Patients with COPD may report that typical respiratory infections are now occurring more frequently, lasting longer, and seeming more severe. Colds bring on shortness of breath, wheezing, and coughing as the most common symptoms (Harding et al., 2022).
SMOKING HISTORY
The key element in taking the history of a patient with COPD is inquiring about smoking. The first symptoms of COPD appear after about 20 pack-years of smoking, and the disease usually becomes clinically significant after 40 pack-years of smoking (Harding et al., 2022).
OTHER IMPORTANT INFORMATION
Besides chronic diseases and heart conditions, other specific problems should be explicitly investigated when taking the history of a patient with COPD:
- Allergy history. Asthma and other allergic syndromes that affect the respiratory system can worsen (or mimic) COPD. There is a great deal of functional and pathologic overlap between asthma and COPD, resulting in asthma–COPD overlap syndrome (Harding et al., 2022).
- Symptoms of clinical depression. Depression is more common in people with chronic illnesses such as COPD. Symptoms of anxiety and depression—such as poor appetite, persistent sadness, inability to focus, restlessness, lethargy, poor self-image, somnolence, suicidal ideation, thoughts of harming self, exhaustion, self-loathing, unexplained weight loss, and insomnia—may be found in the medical history and have been treated effectively by pulmonary rehabilitation (CESD-R, 2024; GOLD, 2025).
Physical Exam
A patient with mild COPD may have no signs of the disease when sitting quietly, and their physical exam may be normal. In contrast, the physical exam of a person with severe COPD can be diagnostic. The physical exam may include measurements of height and body mass as well as spirometry to measure lung function, including forced vital capacity (FVC) and forced expiratory volume (FEV) (GOLD, 2025).
GENERAL APPEARANCE
Patients with emphysematous COPD are typically thin but barrel-chested. They tend to breathe through pursed lips, and they sit leaning forward in a “tripod” position, supporting the upper body on the elbows or the extended arms. This posture widens the chest as much as possible by forcing the diaphragm down and forward.
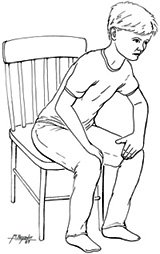
The tripod position. Patient leans forward, resting on elbows or hands, in an effort to expand the chest and ease breathing (McMillen, 2023b). (Source: Jason M. Alexander, MFA. © 2007, Wild Iris Medical Education.)
Patients with chronic bronchitis COPD are typically of normal weight or overweight. They have a productive cough and may be cyanotic. At rest, their rate of respirations is high, often more than 20 breaths per minute. Patients may present as dull and irritable because their state of consciousness can be clouded by hypoxemia.
WEIGHT
The patient’s weight will influence the treatment recommendations. Obesity worsens the symptoms of COPD. On the other hand, many patients with COPD, especially patients with the emphysematous form of COPD, are cachectic and underweight and have wasted muscles. In these cases, nutritional therapy will be important (GOLD, 2025).
CHEST
A patient with COPD with chronic bronchitis but little emphysema may have a normal-sized chest. Significant emphysema, however, leads to a wide, barrel-shaped chest with a flattened diaphragm. In a patient with emphysema, the chest remains perpetually in the position of inhalation. To take a new breath, emphysematous patients must expand their chests beyond the normal position of inhalation. This requires using accessory respiratory muscles of the shoulder, neck, and back (Harding et al., 2022).
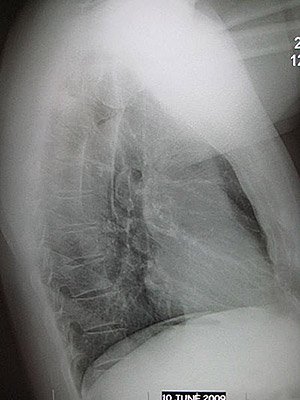
Normal lateral chest X-ray. (Source: James Heilman, MD.)
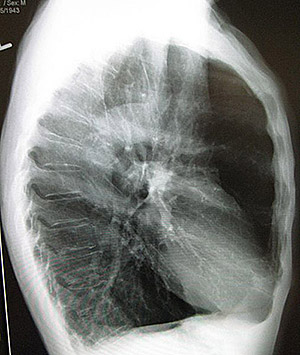
A lateral chest X-ray of a person with emphysema. Note the barrel chest and flat diaphragm. (Source: James Heilman, MD.)
(See also “Chest X-Rays” later in this course.)
LUNGS
The chest of an emphysematous patient is unusually resonant to percussion, and the breath sounds are distant. At the other end of the spectrum, the chest of a chronic bronchitis patient can have dull spots when percussed, and their lungs will be noisy, with rales, rhonchi, and wheezing.
The common feature of all forms of COPD is airway obstruction that worsens as the disease becomes more severe. A simple, direct measure of airway obstruction is the time it takes a patient to exhale an entire lungful of air. A normal person has a forced expiratory time (FET) of <3 seconds. An FET of >4 seconds suggests obstruction, and an FET of >6 seconds indicates considerable airway obstruction at the level of moderate-to-severe COPD.
HEART
COPD can injure the heart in two major ways:
- The chronic inflammatory state of COPD predisposes a person to develop coronary artery disease. Therefore, the history and physical examination of a patient with COPD should look for evidence of ischemic heart problems.
- COPD can cause pulmonary hypertension that strains the right ventricle of the heart. Pulmonary hypertension will intensify the pulmonary component of the second heart sound. In addition, pulmonary hypertension can cause tricuspid valve insufficiency, which can be heard as a holosystolic murmur loudest along the left sternal border. When pulmonary hypertension causes right-sided heart failure (cor pulmonale), the patient will have jugular venous distension and edema of the legs and ankles. Cor pulmonale is a late outcome of COPD and causes a poorer prognosis, although not all people with COPD will develop cor pulmonale.
(Harding et al., 2022)
CASE
Lionel Messenger is a 72-year-old man admitted to the intensive care unit following a myocardial infarction. He has a history of type 2 diabetes mellitus, hypertension, coronary artery disease, hypercholesterolemia, cor pulmonale, and COPD. He presently lies comfortably in bed without pain or difficulty breathing on 2 liters per minute of oxygen by nasal cannula. His cardiac monitor shows sinus tachycardia with a heart rate of 110 beats per minute and occasional premature ventricular contractions (PVCs).
Upon physical examination by the critical care nurse, Mr. Messenger displays clear but diminished breath sounds, a systolic heart murmur, 2+ radial pulses, 1+ pedal pulses, 3+ pitting edema halfway to the knees, jugular vein distension (JVD) while upright, and clubbing of the fingertips. As his condition is stable, he will be transferred to the telemetry unit as soon as a monitored bed is available.
Laboratory Findings
The key chemistry values in a person with COPD are the levels of blood gases (oxygen and carbon dioxide) and the pH of the blood.
BLOOD OXYGEN LEVELS
The severity of a patient’s COPD can be estimated by the degree that the blood gases deviate from normal. In the preliminary stages of the disease, the amount of oxygen in arterial blood is usually within normal limits, measured as its partial pressure (PaO2) (or oxygen tension), with a normal level being 80–100 mmHg.
As COPD worsens, the PaO2 can drop below 60 mmHg. This level signals respiratory distress to the brain, and it strongly activates the respiratory centers. When the PaO2 is below 60 mmHg, a person hyperventilates in an attempt to reverse the hypoxemia by breathing in more air. Unfortunately, hyperventilation due to hypoxemia expels too much carbon dioxide from the bloodstream and causes respiratory alkalosis (a pH imbalance in the blood). Hypoxemia with alkalosis is found in the middle phases of COPD.
In later stages of COPD, the patient does not have the energy to hyperventilate, so carbon dioxide builds up in the blood, with the PaCO2 often reading >50 mmHg. Now the hypoxemia is accompanied by hypercapnia, and the patient develops chronic respiratory acidosis, an ominous sign. Hypoxemia with acidosis is found in the late phase of COPD (Pagana et al., 2022).
Arterial Blood Gases (ABGs)
Early in the course of COPD, ABGs do not need to be checked regularly. However, an early set of baseline values should be taken because they can be used as a comparison to evaluate the degree of change brought on by an acute exacerbation.
| Component | Normal | Critical |
|---|---|---|
| (Harding et al., 2022; Pagana et al., 2022) | ||
| pH | 7.35–7.45 | <7.25 or >7.6 |
| PaO2 (arterial) | 80–100 mmHg | <40 mmHg |
| PaCO2 | 35–45 mmHg | <20 or >60 mmHg |
| HCO – 3 (bicarbonate) | 22–26 | <10 or >40 |
| Base excess (BE) | –2 to +2 mEq/L | ± 3 mEq/L |
| O2 saturation (sat) | 94%–100% | ≤75% |
Pulse Oximetry
Accurately measuring a person’s blood oxygen tension requires drawing arterial blood and testing it in a laboratory. Pulse oximetry is a quicker, noninvasive way to test blood oxygenation. A pulse oximeter has a small probe that can be clipped onto a patient’s finger or earlobe. Using measurements of transmitted light, the oximeter determines the percentage of the patient’s hemoglobin (Hgb) that is saturated with oxygen.
Pulse oximeters are not as accurate as direct oxygen tension measurements from arterial blood gases, and the percentage of hemoglobin saturation measured by an oximeter is not the same as a person’s PaO2. Nonetheless, the two values are related. A person with a normal PaO2 (80–100 mmHg as determined from blood gases) will have a hemoglobin saturation of 94%–100% (as determined by pulse oximetry). A person with hypoxemia of 60 mmHg will have a hemoglobin saturation of approximately 88%. Normal range of oxygen saturation is 94%–100%, but a person with moderate to severe COPD may run lower-than-normal saturation levels when breathing room air. In COPD, dynamic hyperinflation at the end of expiration leads to lower-than-normal oxygen saturation readings, causing exercise intolerance and exertional dyspnea (Harding et al., 2022).
HEMOGLOBIN AND HEMATOCRIT
Routine blood analyses are not needed to manage most cases of COPD. Some people with severe COPD produce excess red blood cells (polycythemia) in response to their chronic hypoxia. This leads to hematocrit (Hct) readings of >52% in men (normal is 43%–52%) and >48% in women (normal is 37%–48%). Another consideration may be that, since oxygen is transported on the hemoglobin (Hgb) molecule, a low Hgb level will cause a decrease in the amount of oxygen available to the tissue.
ALPHA-1 ANTITRYPSIN (AAT) LEVELS
Patients who develop emphysema at an early age (under 40 years) and nonsmokers of any age who develop emphysema are usually tested for their blood levels of the enzyme AAT. (See also “Alpha-1 Antitrypsin (AAT) Deficiency” earlier in this course.) Approximately 3% of all people diagnosed with COPD have an undetected AAT deficiency. The deficiency is diagnosed by a blood level of the protein or AAT phenotype, or by genetic testing. A serum concentration of alpha-1 antitrypsin <15%–20% of the normal value suggests the presence of an AAT deficiency (Harding et al., 2022).
Imaging Studies
COPD is a disease that is defined as having structural and functional abnormalities: COPD causes progressively worsened airflow obstruction in the lungs. Therefore, breathing measurements are better diagnostic indicators of the disease than are static pictures of the lung. Nonetheless, imaging studies play a role in evaluating patients with COPD and their pathologic processes and physiologic consequences.
The most commonly used images for evaluating and managing COPD are chest X-rays and computed tomography (CT) scans. Other modalities that are sometimes used include magnetic resonance imaging (MRI), positron emission tomography (PET), single-photon emission computed tomography (SPECT), electrical impedance tomography (EIT), and optical coherence tomography (OCT).
CHEST X-RAYS
Chest X-rays are used to rule out other causes of airway obstruction, such as mechanical obstruction, tumors, infections, effusions, or interstitial lung diseases. In acute exacerbations of COPD, chest X-rays are used to look for pneumothorax, pneumonia, and atelectasis (collapse of part of a lung).
In its later phases, COPD produces changes that can be seen in chest X-rays:
- When COPD includes significant emphysema, the chest is widened, the diaphragm is flattened, and the lung fields have fainter and fewer vascular markings. Emphysema can make the heart look long, narrow, and vertical, and the air space behind the heart can be enlarged.
- When COPD includes significant chronic bronchitis, chest X-rays have a “dirty” look. There are more vascular markings and more nonspecific bronchial markings, and the walls of the bronchi look thicker than normal when viewed end on. Often, the heart appears enlarged.
CHEST COMPUTED TOMOGRAPHY SCANS
CT scans are now the imaging technique of choice for lung evaluations. Helical or spiral CT scans may be used with contrast medium for better visibility. CT scans, especially high-resolution scans, are better than chest X-rays at resolving the details of the lung abnormalities caused by COPD. Specifically, CT scans can help define which areas of a patient’s lungs are predominately emphysematous and which are predominately bronchiolitic. Late in the disease, high-resolution CT scans are used to evaluate patients with COPD who are to be treated with lung volume reduction surgery (Harding et al., 2022).
Lung Function Tests
Pulmonary function tests (PFTs) are used to assess the extent of a patient’s airway obstruction and include FEV1, FVC, and FEV1/FVC (see below). When COPD is diagnosed, baseline pulmonary function values are recorded. Later tests are used to measure the progression of the disease and to evaluate the effectiveness of treatments.
For COPD, the two general classes of breathing tests are measurements of lung volumes and measurements of airflow rates/volumes. In a patient with COPD, results show a decreased FVC, a decreased FEV1, or a decreased FEV1/FVC combined (Haddad & Sharma, 2023).
LUNG VOLUME
In COPD, airway obstruction makes it difficult to fully empty the lungs. The air that remains keeps the lungs inflated even after a complete exhalation. This makes it more difficult for a patient to pull in sufficient air during the next breath. As a result, the total air volume contained by the lungs increases, but the effective volume of air (the amount of air actually breathed in and out) decreases.
The effective volume of air is called the vital capacity (VC). VC denotes the largest volume of air that can be exhaled after a full inhalation. Usually, this volume is measured by having a patient take as large a breath as possible and then exhale as quickly and forcefully as possible. Forced vital capacity (FVC) is the maximum volume able to be expired after maximal inspiration (Haddad & Sharma, 2023; Harding et al., 2022).
AIRFLOW RATES
Besides limiting the effective volume of air in the lungs, COPD also slows the movement of air inside the lungs. This slowing can be measured directly. Measurements of the rate of air movement during breathing are called spirometric measurements or parameters; more specifically, spirometry measures the volume of air exhaled in a defined period of time (Harding et al., 2022).

A small, handheld spirometry device can be used for quick office or clinic tests. (Source: National Institutes of Health.)
The most common spirometric measurement used for COPD is the 1-second forced expiratory volume (FEV1). This is the maximum amount of air that a patient can breathe out in the first second of a forced exhalation after having taken a full breath.
Spirometry is helpful in evaluating the severity of airflow obstruction in patients with symptomatic COPD. On the other hand, spirometry does not add much to the evaluation of asymptomatic patients with COPD because treatments (other than smoking cessation) are not typically begun until after a patient becomes symptomatic (GOLD, 2025; Harding et al., 2022).
(During the COVID-19 pandemic, the standard method of diagnosing COPD, spirometry, was limited because of the severe amount of coughing and droplet dispersion involved that can spread the COVID-19 virus.)
ANSWERING PATIENT QUESTIONS
Q: What is spirometry?
A: Spirometry measures how much air you breathe and how quickly you can get air into and out of your lungs. Spirometry tests are easy and painless. You breathe forcefully into a tube, and the machine at the other end measures how much air you are moving. Spirometry can detect COPD even before you have many symptoms.
Ranking the Severity of COPD
People with normal lungs can expel most of the air in their lungs within 1–2 seconds. The amount of air forcefully exhaled in the first second (FEV1) is about three quarters of a healthy person’s FVC.
In COPD, airway obstruction restricts the rate of exhaling, and people with COPD cannot exhale a normal amount of air out of their lungs in one second. People with COPD have a FEV1/FVC ratio of <0.70. When a person has an FEV1/FVC ratio of <0.70 and a history of more than 20 pack-years of smoking, they can be given a presumptive diagnosis of COPD (Harding et al., 2022).
The four basic stages of COPD are mild, moderate, severe, and very severe. COPD is staged by the degree to which the FEV1/FVC ratio is <0.70 when corrected for the person’s age, gender, and body build.
| Stage | Severity | Spirometry |
|---|---|---|
| * Predicted FEV1 values adjusted for a person’s age, gender, height, and weight can be calculated from published equations. (GOLD, 2025) |
||
| GOLD 1 | Mild |
|
| GOLD 2 | Moderate |
|
| GOLD 3 | Severe |
|
| GOLD 4 | Very Severe |
|
CASE
Bruce Emerson is a 68-year-old man with a 30-pack-year history of smoking. He quit with the assistance of nicotine replacement therapy (NRT) patches three years ago when he was diagnosed with moderate emphysema-type COPD. He was diagnosed primarily on the basis of symptoms of chronic cough for two years, progressively worse dyspnea at rest, early signs of a barrel chest, less than normal weight, and the following blood gas measurements:
- pH, 7.31
- PaO2, 74
- PaCO2, 48
- Base excess (BE), –3
- HCO – 3 (bicarbonate), 20
- Oxygen saturation (O2 sat), 88%
Mr. Emerson also underwent diagnostic spirometry testing to measure his lung function. At that time, his FEV1/FVC ratio was 0.65 with a FEV1 of 65%, which is indicative of moderate COPD.
Today, Mr. Emerson is undergoing another workup following an extensive period of pulmonary rehabilitation. Pulmonary function retesting shows significant deterioration. His current FEV1/FVC ratio has dropped to 0.55 with an FEV1 of 46%, which is considered severe. His pulmonologist therefore determine that the deterioration of the patient’s lung function necessitates further instruction on exercising as part of his pulmonary rehabilitation program and some changes in his bronchodilator medications.
Differential Diagnoses, Including Asthma
Dyspnea and chronic cough are the presenting symptoms of conditions other than COPD. These conditions include:
- Asthma
- Pneumothorax
- Pulmonary emboli
- Pneumonia
- Lung infections
- Atelectasis
- Interstitial lung disease
- Sarcoidosis
- Effusions
- Upper-airway or foreign-body obstructions
A patient with COPD may also have other comorbidities such as lung masses, respiratory infections, increased incidence of atrial fibrillation, arterial hypertension, heart failure, ischemic heart disease, COVID, bronchiectasis, obstructive sleep apnea and insomnia, periodontitis, GERD, osteoporosis, anemia, polycythemia, anxiety and depression, or cognitive impairment.
Most of these conditions can be identified using imaging studies such as chest X-rays and clinical signs. Anemia or metabolic acidosis can also cause chronic dyspnea, and both of these can be identified by blood studies. A differential diagnosis depends especially on age of onset, chest X-ray (CXR) and CT scan results, volume and character of sputum, and history of smoking (GOLD, 2025).
ASTHMA VS. COPD
Asthma, which is another common obstructive airway disease, is high on the list of differential diagnoses for conditions presenting with both dyspnea and cough. Asthma usually cannot be distinguished from COPD by chest X-rays, clinical signs, or blood studies.
Patients with asthma have hypersensitive airways that are always slightly inflamed, edematous, and filled with immune cells, characteristically eosinophils. Certain inhaled allergens and a variety of stresses can trigger these primed immune cells, causing a flare of the disease (an asthma “attack” or exacerbation) that brings on edema, mucus, and narrowed airways. Like COPD, asthmatic exacerbations will obstruct airways and impede airflow; but unlike COPD, the airway restrictions of an asthmatic exacerbation can be, at least in young people, quickly and almost entirely reversed by bronchodilators.
As people with asthma age, however, their airway obstruction sometimes becomes more fixed and less reversible. Clinically, these people’s disease begins to share more features with COPD, and the two diseases may be hard to distinguish. People with asthma have 12 times the possibility of developing COPD later in life. Determining which disease is present can be important for a patient’s treatment. For example, the dyspnea of asthmatic patients tends to improve markedly when the patient is given steroids, but the chronic dyspnea of most patients with COPD does not improve following steroids (GOLD, 2025).
Some useful distinctions between asthma and COPD include:
- Asthma usually appears in people <30 years of age, while COPD typically appears in people >40 years of age.
- Asthmatic exacerbations are reversed quickly and completely by medications, while the symptoms of COPD are reversed only modestly and temporarily by medications.
- Asthma often runs in families, while COPD usually does not.
| Disorder | Symptoms/Relationship to COPD | Smoking a Factor? |
|---|---|---|
| (Adapted from Harding et al., 2022.) | ||
| COPD |
|
90%–95% with emphysema with no genetic factor |
| Asthma |
|
Symptoms exacerbated with smoking |
| Lung masses |
|
85% |
| Effusions |
|
Possibly |
| Congestive heart failure |
|
Possibly |
| Pneumonia |
|
Possibly |
| COVID-19 |
|
No, cause is infectious |
COPD AND COVID-19 RISK
Most studies have not shown COPD patients to be at a higher risk for becoming infected with the SARS-CoV-2 virus that causes COVID-19. However, having a clinical diagnosis of COPD significantly increases the odds of poor clinical outcomes in patients who contract COVID-19. COPD patients should thus be considered a high-risk group and targeted for preventive measures and aggressive treatment for COVID-19, including vaccination (Tee-Melegrito, 2023).
Patients with COPD are therefore instructed in the importance of protecting themselves from COVID-19 by wearing a mask when indoors or in a crowded situation. However, wearing an N95 mask is not recommended for COPD patients because wearing a tightly fitting N95 may cause additional inspiratory resistance and adversely affect the COPD patient’s respiratory rate, peripheral oxygen saturation, and expired carbon dioxide levels. The standard recommended mask is a triple-layered, surgical-quality mask (GOLD, 2025).
LONG-TERM TREATMENT OF COPD
COPD is a lifelong disease. There is no cure, but it can be treated. COPD requires special medical treatment during acute exacerbations, and after the disease reaches the “moderate” level, it requires daily medications and permanent adjustments to a patient’s lifestyle. GOLD guidelines offer a comprehensive framework for the management of COPD (GOLD, 2025).
The goals of long-term COPD treatments are to:
- Slow the progression of the disease
- Ease the symptoms
- Increase the patient’s ability to be mobile and carry out activities of daily living
- Prevent acute exacerbations
Education is important to improve quality of life and reduce hospital admissions. All patients with COPD should learn about their disease and understand that smoking and air pollution will further damage their lungs. Patients are instructed to make a special effort to avoid respiratory infections and to get yearly influenza vaccinations. In addition to yearly influenza vaccinations, it is recommended that all adults obtain a pneumonia vaccination after reaching age 65. Those at higher risk for pneumonia, such as patients with COPD, are urgently recommended to be vaccinated, often earlier than age 65 (GOLD, 2025).
ANSWERING PATIENT QUESTIONS
Q: What can be done for my COPD?
A: Treatment for COPD helps prevent complications, prolong life, and improve a person’s quality of life. Quitting smoking, staying away from people who are smoking, and avoiding exposure to other lung irritants are the most important ways to reduce your risk of developing COPD or to slow the progress of the disease if you have it.
Treatment for COPD includes medicines such as bronchodilators or steroids. Preventive therapies include flu and pneumococcal vaccines to avoid or to reduce further complications.
As the symptoms of COPD get worse over time, a person may have more difficulty walking and exercising. You should talk to your primary care provider about exercise programs. Ask whether you will benefit from a pulmonary rehab program—a coordinated program of exercise, physical therapy, disease management training, advice on diet, and counseling.
Oxygen treatment and surgery (to remove part of a lung or even to transplant a lung) may be recommended for patients with severe COPD.
At each stage of the disease, there are characteristic medical therapies:
- Mild COPD (GOLD 1) is usually treated with short-acting bronchodilators, which are used as needed for dyspnea.
- Moderate COPD (GOLD 2) requires regular treatments with bronchodilators, sometimes with the addition of inhaled corticosteroids. At this stage, patients are often enrolled in a pulmonary rehabilitation program.
- Severe COPD (GOLD 3) typically requires two or more bronchodilators regularly. Inhaled corticosteroids are added to the regimen to prevent repeated acute exacerbations.
- Very severe COPD (GOLD 4) usually requires the addition of long-term oxygen therapy. Surgical treatments can be appropriate at this stage.

(Source: National Institutes of Health.)
Therapeutic Lifestyle Changes
Medications are the fundamental day-to-day tools for controlling the symptoms of COPD, but there are also effective nonpharmaceutical techniques for treating COPD. These include patient education, smoking cessation, keeping airways clear, and nutritional therapy.
PATIENT EDUCATION / ENERGY CONSERVATION
Self-management interventions attempt to motivate, engage, and coach COPD patients to adopt positive health behaviors and manage their disease better on a day-to-day basis by learning effective skills. Patient education for COPD patients includes:
- Risk factors
- Signs and symptoms of exacerbations
- Compliance with treatment
- Maintaining contact with healthcare providers
- Addressing the complex psychosocial factors of having COPD
COPD patients who use the education given to them to establish healthier self-management behaviors may experience significantly fewer hospital readmissions in the short run (GOLD, 2025).
Following are guidelines a clinician can follow in the area of patient education:
- Teach patients with COPD about their disease. Explain that the disease causes irreversible and progressive problems. Warn patients that they will have episodes in which the symptoms—difficulty breathing, wheezing, productive cough, and tiredness—get worse for days or even weeks.
- Assure patients that they will be helped by medications that make breathing easier. Tell them there are several things they can do to slow the progression of the disease and to lessen the number of acute exacerbations. The most crucial step is to stop smoking. Although smoking has already damaged their lungs, continued smoking will increase the damage and will make their COPD worsen more quickly.
- Let patients with COPD know that they should make every effort to stay active while recognizing the need to monitor and time their efforts throughout the day. In addition, give them practical suggestions that will help them to cope with the inevitable limitations posed by COPD (see box below).
ENERGY CONSERVATION TIPS
- Simplify your tasks and set realistic goals. Do not think you have to do things the same way you have always done them.
- Plan your activities (chores, exercise, and recreation) ahead of time. Space out your activities throughout the day. Do not schedule too many things to do in one day. Do the things that take more energy when you are feeling your best.
- If needed, rest before and after activities.
- If you become tired during an activity, stop and rest. You might need to finish it on another day or when you feel less tired.
- Do not plan activities right after a meal. Rest 20–30 minutes after each meal.
- Ask for help. Divide tasks among family and friends.
- Get a good night’s sleep and elevate your head when sleeping. Be careful not to nap too much during the day or you might not be able to sleep at night.
- Do all of your grooming (shaving, drying your hair, etc.) while sitting.
- If needed, use devices and tools that assist you, such as a walker, shower chair, handheld shower head, bedside commode, or long-handled tools for dressing (such as a dressing stick, shoehorn, or sock donner).
- Wear clothes that have zippers and buttons in the front so you do not have to reach behind you.
- If your doctor says it is okay, you may climb steps. You might need to rest part of the way if you become tired. Try to arrange your activities so you do not have to climb up and down stairs many times during the day.
- Avoid extreme physical activity. Do not push, pull, or lift heavy objects (more than 10 pounds) that require you to strain.
(ALA, 2024d)
SMOKING CESSATION
The cornerstone of management of COPD is smoking cessation. Most patients with COPD have a long smoking history, and many will still be smoking when they are under medical care. From day one, clinicians should strongly urge patients to stop smoking.
Quitting can be difficult since the nicotine in tobacco smoke is powerfully addictive. In addition, the rituals of smoking fill basic psychological needs for many individuals. Therefore, when healthcare professionals merely tell patients to stop smoking, their patients succeed over the long term only 5% of the time. On the other hand, a large study of a smoking cessation program that combines nicotine replacement therapy (NRT) medications with cognitive behavioral therapy (CBT) showed a 50%–70% successful quitting rate (Sandhu et al., 2023).
Healthcare professionals are vital to the success of a smoking cessation program. They may begin by saying to patients: “COPD cannot be cured, but if you continue smoking, the disease will worsen much more quickly. Have you thought about quitting smoking?” Regardless of the patient’s answer, they follow with the offer, “When you’re ready to stop smoking, I’ll be happy to collaborate with you to set up as effective a program as possible.”
Successful smoking intervention programs begin by asking the patient to set a specific quitting date. The programs then maintain continued contact with the patient to provide medication, counseling, support, advice, and a modicum of social pressure. (See also “Resources” at the end of this course.)
ANSWERING PATIENT QUESTIONS
Q: Why should I quit smoking?
A: Statistically, people who stop smoking live longer. If you quit smoking before you are 35, you will live about six years longer. Even if you quit at age 55, you may still add two years to your life. By quitting smoking, you reduce your chances of getting lung disease, heart disease, and cancer. You will feel better and healthier. Smoking injures your senses of taste and smell, and quitting smoking will even make food taste better.
Q: I like to smoke, and I know people who have lived a long time even though they were smokers. Why should I go through the agony of stopping something I enjoy? Besides, I may not even be able to quit.
A: Cigarettes are legal addictive drugs, and they are easier to buy and less expensive than illegal drugs—but smoking is gambling, with bad odds. As a smoker, you have a 1-in-3 chance of dying earlier than you would if you quit. When you do die, it will most likely be of heart disease, stroke, cancer, or COPD. Smoking is responsible for about 1 out of every 5 deaths in the United States, and almost half a million Americans die each year from diseases caused by smoking.
Your smoking can also hurt the people around you. Breathing in another person’s smoke can cause lung problems in children and cancer and heart disease in adults. Pregnant women and new parents can protect their baby’s health by stopping smoking now.
Sure, it is tough to quit smoking. Staying healthy and protecting the health of the people around you is difficult. But it’s important not to hide behind the excuse that you won’t be able to stop smoking. Studies suggest that most people can quit smoking with the combination of behavioral and pharmacologic therapy.
Q: What is the first thing I need to do once I’ve decided I want to quit?
A: You should set a quit date. Then make an appointment to see your primary care provider before the quit date. They will help you devise a plan that will make quitting easier.
Also, plan to join a support group or a stop-smoking program. The American Lung Association has an online stop-smoking program called “Freedom from Smoking Online.” Another helpful organization is Nicotine Anonymous, which runs 12-step programs with group support. (See also “Resources” at the end of this course.)
Here are some other general tips:
- Pick a suitable time to quit, a time when you won’t be under a lot of stress.
- Face the fact that it may not be easy and that you may have uncomfortable symptoms for a few weeks. You may get headaches or be sleepy or dizzy. You may become irritable or nervous. You will probably have cravings for a cigarette.
- Add some extra exercise to your quitting program. Walking, for example, is a great stress reducer.
- Tell your friends and family you are trying to quit smoking. Get their help to distract you, to keep up your spirits, and to be there when you need to complain.
Pharmacologic Therapy for Smoking Cessation
The pharmacologic aspect of smoking cessation programs attempts to ease the effects of nicotine withdrawal. Smokers who need their first cigarette within a half hour of getting up in the morning are likely to be highly addicted to nicotine. When these people stop smoking, they become anxious, irritable, easily angered, easily tired, and depressed. Their sleep is disrupted. They have difficulty concentrating. These withdrawal effects are common during the first 2–3 weeks after quitting.
FDA-approved medications for the treatment of tobacco use include:
- Nicotine gum
- Nicotine inhaler
- Nicotine lozenge
- Nicotine nasal spray
- Nicotine patch
- Bupropion SR (Wellbutrin, Zyban)
- Varenicline (Chantix)
(McCuistion et al., 2022)
Nicotine replacement therapy (NRT). To lessen withdrawal symptoms, nicotine can be taken in low doses without smoking to relieve the symptoms of craving. The transdermal patch is the only form of NRT that secretes a continuous dose of nicotine to prevent cravings. Other forms of nicotine replacement include gum, lozenges, inhalers, and nasal sprays; these should be used on an as-needed (PRN) basis for cigarette cravings for about two weeks, and then the doses should be tapered. The gum, lozenges, and inhaler help to satisfy oral cravings, and the inhaler raises nicotine blood levels more rapidly than the other routes of administration.
Nicotine patches are marketed as Habitrol and NicoDerm CQ; nicotine gum includes Nicorette. Prescription nicotine replacement therapy is available only under the brand name Nicotrol, available both as a nasal spray and an oral inhaler.
Nicotine replacement products can cause the following side effects:
- Nausea
- Dizziness
- Weakness, vomiting, fast or irregular heartbeat, mouth problems (with the lozenge or gum)
- Redness or swelling of the skin around the patch that does not go away when the patch is removed
If these side effects occur, the patient is instructed to discontinue the product’s use and report the effects to their primary care provider (McCuistion et al., 2023).
Since nicotine is a vasoconstrictor, nicotine replacement therapy is contraindicated in people with coronary artery disease (CAD). Vasoconstriction causes increased arterial resistance that may result in an increase in blood pressure. Additionally, individuals with CAD with increased vasoconstriction may experience a reduction in the diameter of the coronary arteries. This can lead to angina or myocardial infarction. In the hospital setting, patients with CAD who are experiencing nicotine withdrawal are given sedatives to reduce symptoms rather than nicotine replacement medications for these reasons.
Antidepressants. The FDA has approved the antidepressant bupropion SR (sustained-release) to help patients for whom nicotine replacement therapy has not worked. Bupropion raises levels of dopamine in the brain, which helps to relieve nicotine cravings. Treatment usually lasts for 7–12 weeks but may be continued for the needed therapeutic effect. It should not be stopped abruptly; patients should be weaned off slowly to avoid withdrawal symptoms.
Nicotine agonists. The FDA has approved varenicline (Chantix), a nicotine agonist, for antismoking therapy. Varenicline binds to nicotine receptors and prevents nicotine from activating the receptors, while producing a smaller stimulant effect than nicotine. Since varenicline contains no nicotine, it does not cause vasoconstriction that can reduce blood flow to the myocardium, making it the drug of choice for patients with a cardiac history. It also stimulates the release of dopamine. Side effects may include nausea and vomiting, insomnia, headache, abnormal dreams, constipation, diarrhea, fatigue, malaise, upper respiratory tract infection, dyspnea, chest pain, abdominal pain, xerostomia, appetite changes, rash, or emotional disturbances.
Electronic cigarettes (or e-cigarettes) contain vaporized liquid nicotine and are believed to aid in smoking cessation by reducing the amount of nicotine used, but there is not yet sufficient research to support this. No prescription is needed (McCuistion et al., 2023).
ANSWERING PATIENT QUESTIONS
Q: What medicines should I take when I’m trying to stop smoking?
A: There are a variety of antismoking medicines, and your primary care provider can suggest the best one for you. Nicotine is an addictive drug. For many people, nicotine replacements help to keep withdrawal symptoms to a minimum. Nicotine replacements come as patches, gums, lozenges, and an inhaler. Your primary care provider can also prescribe a nicotine-free tablet called Chantix, which reduces withdrawal symptoms. Some people get help from the antidepressant bupropion, which is a prescription medicine.
Q: Aren’t nicotine replacement products just as bad as smoking?
A: No, nicotine replacements do not have all the tars and poisonous gases that are found in cigarettes. Furthermore, these medicines give you less nicotine than a smoker gets from cigarettes. Nicotine replacement products (patches, gums, lozenges, or inhalers) should not be used by people who are pregnant or nursing. People with other medical conditions should check with their primary care provider before using any nicotine replacement product. It is important that smokers quit smoking completely before starting to use nicotine replacements.
CASE
Elsa Hunter is a patient with moderate COPD who is struggling with quitting smoking. She has tried nicotine patches and gum, the nicotine agonist Chantix (varenicline), hypnotherapy, acupuncture, and counseling. Each method has been temporarily successful, and then Ms. Hunter has started smoking again. Ms. Hunter states that the patches caused skin irritation and scarring, the gum didn’t work, Chantix caused severe nausea, and the hypnotherapist “couldn’t put her under.”
Although Ms. Hunter understands the dangers of smoking and the effects on her health, she continues to return to smoking in times of stress. The nurse sits with Ms. Hunter to discuss the types of stressors that trigger her addiction and some strategies to avoid them or to manage them in other ways. The physician also orders the antidepressant Zyban (bupropion) to reduce nicotine cravings.
At her next follow-up visit, Ms. Hunter informs the nurse that she has been smoke free for the past six months “thanks to the Zyban.”
KEEPING AIRWAYS CLEAR
Patients with COPD with significant chronic bronchitis must keep their airways clear. They should be encouraged to cough up sputum, and they should not get in the habit of using cough suppressants or sedatives. Postural drainage can help patients who cannot clear their secretions by coughing. This is a technique that patients can be taught to employ at home in which they place themselves in a variety of body positions that encourage gravity-assisted drainage of the lungs.
Most people’s lungs secrete extra mucus in response to inhaled irritants. To avoid stimulating excess secretions, patients with COPD are advised to stay out of smoke-filled rooms and to stay indoors during air pollution alerts. Home air conditioners and air filters are effective at keeping indoor air clear of particulates.
ANSWERING PATIENT QUESTIONS
Q: I have COPD. What can I do at home to keep the air clean?
A: It is important to keep the air in your home clean. Keep your windows closed and stay indoors when there is a lot of pollution or dust outdoors. When you cook, keep smoke and cooking vapors out of the air by using an exhaust fan or opening a door or a window. Don’t let anyone smoke in your house. Unless your fireplace is the only way for you to heat your home, you should not burn wood or kerosene in your home. Avoid using any aerosol (spray) products. Don’t use strong perfumes. When your house is being painted or sprayed for insects, stay away as long as you can or until the fumes have disappeared.
NUTRITIONAL THERAPY
The symptoms of COPD improve when patients who are overweight lose weight. Some patients with COPD, however, have the opposite problem: they have become thin and malnourished. Weight loss is a predictor of more exacerbations and a poorer prognosis.
Weight loss results in part from the high energy cost of breathing with COPD. In addition, the chronic inflammatory state underlying COPD tends to put the body’s metabolism into a catabolic state, in which larger molecules such as tissue are broken down into smaller molecules. This constant breakdown of tissue increases the body’s metabolic rate, causing further weight loss. Other factors that may contribute to malnutrition are poor appetite, altered taste due to mouth breathing, sputum, fatigue, anxiety, depression, infections, and side effects of medications (Harding et al., 2022).
To help maintain a healthy body weight, malnourished patients with COPD are given dietary counseling that includes specific recommendations for meals and meal supplements that are nutritionally balanced and that contain sufficient calories to make up for the work of breathing. Additional calories are also recommended to balance the calories burned during the exercise training recommended for people with COPD (GOLD, 2025).
Suggestions for decreasing dyspnea and conserving energy to lower the number of calories burned include:
- Rest and avoid nebulizer treatments, if possible, one hour before and after meals.
- Use a bronchodilator (inhaler) before meals.
- Use an oxygen nasal cannula during meals if oxygen is needed while eating.
- Have broken or missing teeth fixed to make eating easier.
- Walk or get out of bed for meals to promote appetite.
Patients can also be taught to increase calorie and protein intake without having to increase the amount of food they eat. An additional 25–45 kcal per kg of body weight and 1.5 grams of protein per kg of body weight may be needed to maintain body weight. The malnourished COPD patient may need up to 2.5 grams of protein per kg of body weight to restore muscle mass.
Suggestions for improving weight gain include:
- Eat higher-calorie foods first.
- Limit liquids at mealtimes so as not to fill up on them.
- Eat meals more frequently (five or six per day), and have snacks between meals.
- Add margarine, butter, sauces, mayonnaise, gravies, and peanut butter to food.
- Keep favorite foods and snacks available.
- Eat cold foods (which cause less of a feeling of fullness than hot foods).
- Keep prepared meals available for times when too fatigued or short of breath to prepare meals or snacks.
- Eat larger meals when not as tired.
- Avoid gassy foods (e.g., cabbage, beans, cauliflower).
- Add skim milk powder (2 tbsp) to regular milk for additional calories and protein.
- Use milk and half-and-half instead of water when making soups, puddings, and cocoa.
- Add grated cheeses to sauces, casseroles, soups, potatoes, sandwiches, and vegetables.
- Choose desserts that contain eggs (custards, sponge cake, angel food cakes, bread pudding, rice pudding, and smoothies).
(Harding et al., 2022)
Pulmonary Rehabilitation and Integrated Care
Pulmonary rehabilitation (PR) is the term for a comprehensive, evidence-based, multidisciplinary program designed to assist patients with COPD who are having difficulty with breathing and activities of daily living (ADLs). A self-management COPD program is structured, personalized, and aimed at motivating and supporting patients to adopt positive health practices and develop skills to better manage their illness.
The PR process uses collaborations between COPD patients and healthcare personnel. Most PR programs involve a respiratory therapist, occupational therapist, physical therapist, and dietitian. Physicians, pharmacists, and nurses may also be involved, but not at every meeting with the patient. PR programs are delivered in inpatient, outpatient, clinic, physician office, telehealth, and home settings. A multidisciplinary and multifaceted pulmonary rehabilitation program has been shown to reduce the number of hospital readmissions for a three-month period for COPD patients (GOLD, 2025).
PR programs include assessment, exercise therapy, education, and psychological support. The benefits are maximization of functional status and the reduction of healthcare costs by promoting self-management of symptoms. Some rehabilitation programs continue for an extended time, but most run for a few weeks and then give patients individualized instructions for continuing at home.
The primary objective of a PR program is to restore individual patients to as independent a level of function as possible with an improved health-related quality of life. Evidence has shown that dyspnea symptoms improve in patients with COPD who undergo a PR regime. PR is proven to be a cost-effective treatment model and reduces the number of hospital admissions, but it cannot be substantiated that PR extends the life of patients with COPD (Harding et al., 2022).
EDUCATION IN PULMONARY REHABILITATION
Patient and family education are central to all PR programs. In these education sessions, patients and their families learn details about COPD and its treatment. Education informs the patient and family in how to self-manage the disease in collaboration with the various PR disciplines. Education topics may include understanding chronic lung disease, medications, breathing control, oxygen therapy, heart health, falls prevention, diagnostic tests, and advance care planning. Increasing patient knowledge leads to essential behavior changes, although education alone does not improve outcomes (GOLD, 2025).
ANSWERING PATIENT QUESTIONS
Q: What is pulmonary rehab?
A: Pulmonary rehabilitation is a program that includes regular exercise, training in how to manage your disease, and practical advice, all of which help you to stay active and remain able to conduct your day-to-day activities. After some medical breathing evaluations, you will meet with a pulmonary rehab team and make a plan that is best for your disease and your lifestyle. Usually, there are meetings, exercise classes, suggestions for long-term improvements in your lifestyle, and an advisor whom you can always contact for advice.
ASSESSMENT TESTING
COPD assessment testing measures several parameters (cough, phlegm, chest tightness, breathlessness, limited activities, confidence leaving home, sleeplessness, and energy) to determine a COPD patient’s degree of symptom severity and exacerbation risk. Several other assessment tests can be used to evaluate COPD patients for their ability to tolerate exercises and endurance training.
The 6-minute walk test (6MWT) is well-known endurance field test used to predict a COPD patient’s tolerance for endurance training through exercise. Another widely used COPD patient assessment tool is the Modified Medical Research Council Questionnaire (mMRC). This is a simple, five-question tool that measures the degree of breathlessness. It is also considered a predictor of future mortality risk (Richards, 2024). (See also “Dyspnea and Its Spiraling Effects” earlier in this course.)
The most commonly used tests to assess balance in COPD and other chronically ill patients are the Berg Balance Scale (BBS) and the Balance Evaluation Systems Test (BEST). The BEST test can also be modified and administered as the Mini-BEST test and the Brief-BEST test for patients who are unable to tolerate the complete BEST test because of muscle weakness, pain, or dyspnea. These tests can also be used after six weeks of an intervention of balance training to assess whether or not the training has been effective (Awell, 2024; Cleveland Clinic, 2024).
PROGRAM PLANNING
PR programs are tailored to the needs of each individual. The number of sessions per week, need for nutritional counseling, need for psychological support, and need for oxygen supplementation are determined after a careful assessment of each patient’s capabilities and desired goals. The best possible benefits occur in a PR program lasting 6–8 weeks. There is no evidence of any additional benefits from expanding the program to 12 weeks’ duration. The types of exercise modalities are specifically individualized regarding the duration of each exercise, the number of repetitions, the number of weights, and the progression for increasing exercise (GOLD, 2025).
EXERCISE TRAINING
A goal of PR is to optimize the functional status of a patient with COPD by exercise training and collaborative self-management. Exercise training supervised by occupational and physical therapists does not improve lung functioning, but it can reduce COPD symptoms and increase the amount of exercise that the patients can do without being stopped by dyspnea. It can also reduce the number of hospitalizations for acute exacerbations.
Physical inactivity is the greatest source of the muscle weakness that affects patients with COPD, causing exercise intolerance and the wasting of skeletal and respiratory muscles. Although people with COPD have irreversible breathing difficulties, exercise training—including interval training, strength training, upper- and lower-limb training, and transcutaneous neuromuscular electrical stimulation—can significantly increase a patient’s strength and endurance and reduce their fatigability. These improvements result from increased muscle size (specifically, cross-sectional area), increased blood flow to muscles, increased oxidative enzyme capacity, and reduction of lactic acid production during exercise (ALA, 2023b).
Endurance training improves exercise-induced hyperinflation, exertional dyspnea, heart-rate recovery, and muscle dysfunction in COPD. Endurance training in the form of walking and cycling retards the progression of activity intolerance in patients with COPD, as do unsupported upper extremity exercises, such as cross-body weight-lifting.
Typical PR programs include graded aerobic exercises, such as regular sessions of walking or stationary bicycling three times weekly. The walking exercise program, for example, might begin with slow treadmill walking for only a few minutes. Gradually, the length and speed of the walking is increased. The goal would be for the patient to walk in gradually increased increments without needing to stop because of shortness of breath. At that point, the patient would be assigned a maintenance exercise program to be done at home, which may include stretching, weight training, and a stationary bicycle.
Strength training can improve not only muscle strength and quality of life but also exercise capacity in patients with COPD. Strength training may utilize either free weights (such as dumbbells) or weight machines. For a patient starting out with strength training, a repetition range of 8–12 is appropriate. The training frequency should be 2–3 days per week.
Balance training can improve endurance and exercise capacity. COPD eventually causes skeletal muscle weakness, slow gait, and decreased physical activity ability secondary to dyspnea and fatigue from the overuse of respiratory muscles. Balance impairment is directly connected to falls, and research suggests that specifically improving balance control can result in significant improvement in the ability to perform ADLs more independently (Tounsi et al., 2021).
Supplemental oxygen is recommended during exercise for patients who experience severe exercise-induced hypoxemia. Oxygen saturation may need to be monitored in patients whose oxygen dependency may otherwise prevent them from exercising. Increased flow rates may enable oxygen-dependent patients to exercise longer and with less dyspnea. This may improve exercise endurance during a high-intensity exercise program (ALA, 2023a).
BREATHING INSTRUCTION
Rehabilitation sessions also include breathing instruction that teaches patients how to slow their rate of breathing by pursing their lips and how to rest the upper respiratory muscles by using abdominal breathing instead of chest breathing.
PURSED-LIP BREATHING
This technique can be used during the difficult part of any activity, such as bending, lifting, or stair climbing. It should be practiced four or five times a day to establish the correct breathing pattern. Pulmonary rehabilitation professionals can teach this technique by demonstrating the following steps:
- Relax the neck and shoulder muscles.
- Inhale slowly through the nose, as if you are smelling something, for 2 seconds.
- Pucker or “purse” the lips like you’re blowing out the candles on a birthday cake.
- Exhale slowly and gently through pursed lips for 4–6 seconds.
- Repeat the steps, if needed.
Pursed-lip breathing helps the individual to:
- Focus on breathing
- Breathe slowly and deeply
- Release trapped air from the lungs
- Decrease feelings of breathlessness
- Increase the amount of time one can exercise or perform an activity
- Breathe in oxygen and exhale carbon dioxide more effectively
(COPD Foundation, 2024)
NEUROMUSCULAR ELECTRICAL STIMULATION (NMES)
Some patients with COPD have such poor lung function or such weak musculature that they cannot take part in the usual aerobic exercise training programs. NMES has shown improvement in patients’ physical activity, exercise tolerance, quality of life, and survival by helping to prevent and improve physical deconditioning. This is an individualized protocol in which the intensity (amplitude), frequency, duration, and waveform of the stimulus to the nerves is determined for each patient, causing the muscles to contract. Precise patient and family education is needed to ensure the device is properly used.
NMES may increase muscle strength, exercise capacity, and standing balance, and may reduce dyspnea of outpatients with severe COPD. NMES routines are safe for most patients, are inexpensive, and can be done at home. They can be used during acute COPD exacerbations and with bedridden patients (LoMauro & Gervasoni, 2024).
PSYCHOLOGICAL SUPPORT
Multiple studies have proven there is a direct correlation between reduction of measurable anxiety and depression levels in patients with COPD when they participate in PR. Therefore, PR is recommended as a viable adjunct to other treatments for patients with COPD at any stage of the severity of the disease. The degree of psychological improvement brought about by the program warrants PR being included in treatment of anxiety and depression in this population (GOLD, 2025). Clinical depression as a comorbidity with COPD is associated especially with patients who are younger, female, and smokers, and with a lower FEV1, cough, history of cardiovascular disease, and poor prognosis.
ACTIVITIES OF DAILY LIVING
Occupational therapists (OTs) in particular play a key role in teaching patients with COPD to maintain their independence and abilities in performing necessary ADLs without developing activity intolerance. OTs are involved in approximately 30% of all individual pulmonary rehabilitation programs.
Screening tools for ADLs that may be used in PR settings include:
- Chronic Respiratory Disease Questionnaire-Dyspnea (CRQ-Dyspnea) is used to measure perceived dyspnea on a seven-point Likert scale in the problem or difficulty areas corresponding with the COPM occupations (see below). Higher scores indicate less breathlessness in COPD patients (ALA, 2023b).
- Barthel Index (BI) measures performance in feeding, bathing, grooming, dressing, bowel control, bladder control, toileting, chair transfer, ambulation, and stair climbing, and is one of the most commonly used assessments of functional independence. Based on scoring of up to 100 points, a higher score indicates greater independence (MDCALC, 2024).
- Canadian Occupational Performance Measure (COPM) helps patients to self-assess performance, prioritize what needs to be addressed, and identify intervention goals to improve aspects of self-care, leisure, and productivity. It is the most commonly used occupational therapy outcome measure and is used in 50 countries (CAOT, 2024).
Strategies are then introduced in order to reduce effort associated with daily routines and to reduce fatigue, shortness of breath, and the work of breathing due to the workload and symptom burden of ADLs. These may involve altering body mechanics or altering the environment. Adaptive equipment recommendations may include items such as a long-handled shoe or sock aid, and durable medical equipment suggestions may include a tub seat for seated showers.
Energy conservation techniques include making larger meals to freeze, letting dishes air dry, grouping task items together to minimize unnecessary searches, sliding rather than carrying items, shopping with someone who can carry grocery bags, or using grocery home delivery services (Koolen et al., 2021; Yamaguchi et al., 2021).
CASE
Stuart Moody is being discharged from the hospital with a new diagnosis of mild to moderate emphysema-type COPD. His physician has ordered that Mr. Moody start a pulmonary rehabilitation program with a follow-up visit in six weeks. The nurse explains to Mr. Moody that a PR program consists of education about exercise training, nutritional counseling, medications, how to self-measure peak flows, breathing training, panic control, and airway control.
Mr. Moody will see a registered dietitian in consultation before discharge to help him maintain or achieve his correct weight. He will also collaborate with a physical therapist to learn progressive aerobic, strengthening, and resistance exercises, and an occupational therapist to learn new body mechanics for easier performance of ADLs.
Mr. Moody is fortunate to be started on PR at this time. Previously, PR was only ordered for moderate-to-severe COPD. He is also fortunate that Medicare now covers PR.
Pharmacologic Therapies
The medicines currently available for COPD focus on long-term therapy with the goal of preventing symptoms, reducing the frequency and severity of exacerbations, improving exercise tolerance, and improving general health. Stepped therapy—the process by which additional medications are added as symptoms progress—is the standard in treating COPD. Inhaled and systemic drugs for patients with COPD include:
- Beta-adrenergic agents
- Cholinergic antagonists
- Methylxanthines
- Corticosteroids
- Nonsteroidal anti-inflammatory drugs (NSAIDs)
- Phosphodiesterase inhibitors (in severe cases)
- Mucolytic agents
Drug therapy is used to reduce the extent to which dyspnea restricts a patient’s activities. Most COPD drugs work by keeping airways as wide open as possible. Medications (bronchodilators) used to reduce airflow obstruction are not typically given to asymptomatic patients with COPD (Healthline, 2024).
BRONCHODILATORS
Bronchodilators are the “workhorses” of the COPD medications. Although spirometry shows that bronchodilators only modestly reduce airway obstruction in most patients with COPD, regular doses of bronchodilators relieve dyspnea sufficiently for patients with COPD to increase their levels of activity. Combination treatment increases FEV1 and reduces symptoms and exacerbations more effectively than monotherapy.
Bronchodilators relax the smooth muscles in the walls of the lungs’ airways. This widens the airways and allows air to move through them more easily. Short- and fast-acting bronchodilators are used as “rescue” medicines to relieve sudden bouts of dyspnea and coughing. Long-acting bronchodilators are used in daily, regularly scheduled drug regimens to prevent airway obstruction. Bronchodilators prevent hyperinflation of the lungs at rest and during exercise and thereby improve exercise intolerance.
All symptomatic patients are prescribed a short-acting rescue bronchodilator that they can use to recover from a bout of suddenly worsening dyspnea. Either short-acting parasympatholytic or short-acting sympathomimetic bronchodilators can be used as fast-relief medications (GOLD, 2025; Harding et al., 2022).
Parasympatholytic Bronchodilators
The parasympatholytic bronchodilators are anticholinergic drugs, which relax the smooth muscles in the airway by blocking the effect of acetylcholine, produced by the parasympathetic nervous system.
The most commonly prescribed short-acting anticholinergic bronchodilator is ipratropium (Atrovent). Ipratropium is relatively inexpensive and widely available. It is usually administered via a metered-dose inhaler (MDI), although there are other formulations. It can be used as a PRN medication. It takes effect in 15–30 minutes, has its peak action in 1–2 hours, and lasts 4–6 hours.
Traditionally, ipratropium has also been used as the main anticholinergic in long-term drug regimens. However, studies have shown that tiotropium (Spiriva) is a more effective drug. Tiotropium is a longer-acting anticholinergic bronchodilator. It is more expensive than ipratropium, but a typical dose lasts an entire day. Tiotropium is helpful when used alone and is even more effective in combination with a long-acting beta agonist. Tiotropium is inhaled as a powder via a dry powder inhaler (DPI) (GOLD, 2025; Harding et al., 2022).
When used correctly, MDIs and DPIs deliver the medication directly to the airway. MDIs work best when connected to a “spacer” to guide the medication down the patient’s airway.
HOW TO USE A METERED-DOSE INHALER WITH A SPACER

Metered-dose inhaler. (Source: National Institute of Allergy and Infectious Diseases.)
Clinicians can provide these instructions to educate patients with COPD on using their MDI:
- Make sure that the metal canister of the MDI is inserted correctly into the plastic “boot” or holder.
- Remove the cap from the mouthpiece of both the MDI and the spacer.
- Insert the MDI mouthpiece into the soft opening of the spacer. The MDI canister must be in an upright position.
- Shake the MDI with attached spacer several times.
- Breathe out, away from the spacer, to the end of your normal breath.
- Place the mouthpiece of the spacer into your mouth, past your teeth, and above your tongue. Close your lips around the mouthpiece. If you are using a spacer with a mask, place the mask over your nose and mouth. Be sure the mask has a good seal against your cheeks and chin; there should be no space between the mask and your skin.
- Press down on the top of the metal canister once to release the medicine into the spacer.
- Breathe in deeply and slowly through your mouth. If the spacer makes a “whistling” sound, you are breathing in too quickly. You should not hear a whistle.
- Hold your breath for 5–10 seconds.
- Breathe out slowly.
- If you are instructed to take more than one puff (spray), wait about 15–30 seconds, or 1 minute for beta-2 agonists (or as directed by the package insert), before taking the next puff. Then repeat steps 4 through 10.
- Replace the cap on the mouthpiece of the MDI inhaler and spacer after you have finished.
- If you are inhaling a steroid, rinse your mouth out with water, swish, and spit out the water.
(Harding et al., 2022)
HOW TO USE A DRY POWDER INHALER

Dry powder inhalers (left to right): Turbuhaler, Accuhaler, Ellipta devices. (Source: National Institute of Allergy and Infectious Diseases.)
Clinicians can provide these instructions to educate patients with COPD on using their DPI:
- Remove all candy, food, or gum from your mouth.
- Stand up straight.
- Hold the inhaler level to the floor.
- Open the inhaler, with the mouthpiece facing you.
- Slide the lever away from you until you hear it click. This means the medicine has been released. Be careful not to tip the inhaler or slide the lever again; if you do, the medicine will fall out and be wasted.
- Take a deep breath in and then breathe out, exhaling as much air from your lungs as you can. Do not breathe out into your inhaler because this could affect the dose.
- Place the inhaler in your mouth, seal your lips tightly around it, and take a quick, deep breath in. This will ensure that the medicine moves down deeply into your lungs. You may not taste or sense the medicine going into your lungs.
- Hold your breath for 10 seconds or as long as you can to disperse the medication into your lungs, and then breathe out.
(Harding et al., 2022)
Sympathomimetic Bronchodilators
Beta-2 adrenergic agonists are a class of sympathomimetic bronchodilators that act by mimicking the effect of norepinephrine on airway muscles, causing smooth muscles to relax, thereby widening the airways. Muscle tremors, tachycardia, and heart palpitations are the most common side effects of beta-2 agonists, but when the medicines are inhaled (as opposed to taken in oral formulations), the side effects are usually mild.
Short-acting beta-2 agonists are the most commonly prescribed sympathomimetic bronchodilators and include albuterol (Accuneb, ProAir, Proventil, and Ventolin) and metaproterenol (Alupent). These drugs are usually administered via either MDI or DPI. Short-acting beta-2 agonists such as albuterol and metaproterenol take effect in 5–15 minutes and last for 2–4 hours.
Short-acting beta-2 agonists are used as rescue medicines when a patient needs immediate relief from sudden episodes of increased dyspnea. A short-acting beta-2 agonist can also be added to an anticholinergic drug as part of a regular drug regimen.
Long-acting beta-2 agonist bronchodilators include formoterol (Foradil) and salmeterol (Serevent). These drugs are more expensive than albuterol or metaproterenol, but a typical dose lasts for at least 12 hours. Inhalation is the recommended route for administering the long-acting beta-2 agonists (GOLD, 2025; Harding et al., 2022).
Phosphodiesterase inhibitors, or xanthines, are another class of sympathomimetic bronchodilators. They act by stimulating the release of norepinephrine, which then relaxes smooth muscles in the airways of the lung.
Previously, the phosphodiesterase inhibitor theophylline (Elixophyllin, Theo-Dur) was used to dilate airways, stimulate the respiratory centers of the brain, and improve the function of respiratory muscles. Due to its side effects (diarrhea, nausea, poor appetite, weight loss, abdominal pain, headache, sleep disturbances, and cardiac dysrhythmias) and lower efficacy, theophylline is now rarely used.
Two newer phosphodiesterase inhibitors, cilomilast (Ariflo) and roflumilast (Daxas, Daliresp), appear to be safer than theophylline. Roflumilast is used only for severe exacerbations (GOLD, 2025; Harding et al., 2022).
Bronchodilator Regimens
Patients vary in their response to bronchodilators, so the most effective drug regimens are those that have been individually tailored. Finding the right drug or set of drugs is empirical. Patients may try many different inhalers until finding a combination that works best for them. When drug combinations are being tried, it is best to introduce the drugs one at a time to learn the patient’s response to that drug only.
For patients with chronic stable COPD, short-acting bronchodilators will eventually be insufficient to control their symptoms. Currently, the long-acting anticholinergic drug tiotropium is usually recommended as the first drug to try in a regular daily medication regimen. It is taken once daily and it does not have the side effects of sympathomimetic drugs, but it is generally not as effective as the beta-2 agonists and is recommended only if the COPD patient cannot tolerate the beta-2 agonist side effects.
Concurrently, a short-acting beta-2 agonist, such as albuterol, is usually prescribed as a rescue drug. If this initial regimen is insufficient, the short-acting beta-2 agonist is added to the regularly scheduled drug regimen rather than being used only when needed. The combination of ipratropium and albuterol is available commercially (DuoNeb) as an inhalant.
As COPD progresses, most patients do better with combinations of two or three bronchodilators. In American and Western European medicine, theophylline (or another phosphodiesterase inhibitor) is usually the last bronchodilator to be added.
If they are to be followed faithfully, drug regimens must be realistic. Bronchodilator therapy with two or three drugs is expensive. In addition, using inhalers can be physically difficult for some people, especially older adults, and physicians may need to modify an optimal pharmacologic therapy to make it practical for a particular patient (GOLD, 2025; Harding et al., 2022).
BRONCHODILATORS
Short-acting beta agonists
- Action: relaxes bronchiolar smooth muscle
- Drugs: albuterol (Proventil), metaproterenol (Alupent), erbutenol (Maxair), terbutaline (Brethine)
- Side effects: tachycardia, palpitations, anxiety, muscle tremors
- Comments: fast-acting rescue drug; taken five minutes before other inhalers; used for treatment
Long-acting beta agonists
- Action: relaxes bronchiolar smooth muscle
- Drugs: salmeterol (Serevent)
- Side effects: tachycardia, palpitations, anxiety, muscle tremors
- Comments: not to be used for acute onset of symptoms; inhaler must be shaken since drug separates; used for prevention
Anticholinergic agents
- Action: inhibits parasympathetic nervous system
- Drugs: ipratropium (Atrovent)
- Side effects: cough, dry mouth
- Comments: may exacerbate cardiac symptoms; inhaler must be shaken since drug separates; used for prevention; must be carried at all times if used as a rescue drug
(GOLD, 2025; Harding et al., 2022)
CORTICOSTEROIDS
Corticosteroids, also called glucocorticoids, are “two-edged swords.” They are effective anti-inflammatory medicines used to reduce the inflammatory response that underlies or exacerbates many diseases. However, the continued use of corticosteroids may cause Cushing’s syndrome, glaucoma, cataracts, myopathy, ulcers, osteoporosis, hyperglycemia, poor wound healing, and the inability to overcome infections.
In stable COPD, the problems that result from the long-term use of oral or systemic corticosteroids usually outweigh the drugs’ benefits. Inhaled steroids—such as fluticasone (Flovent), beclomethasone (Beclovent, Beconase), and budesonide (Pulmicort Turbuhaler)—have fewer adverse effects than oral formulations, and approximately 10% of people with COPD find that regularly inhaled steroids reduce their airway obstruction. For this population of patients, inhaled steroids can be a useful addition to the other regularly scheduled bronchodilators.
The regular use of inhaled corticosteroids is usually reserved for patients with severe COPD. In people with severe COPD, steroids will reduce the number of exacerbations and the rate of mortality. For people with severe COPD, inhaled corticosteroids are typically combined with a long-acting beta-2 agonist in a regular treatment regimen. Regular use of inhaled corticosteroids for COPD, however, also increases a patient’s risk of developing pneumonia.
The usefulness of corticosteroid therapy cannot be predicted in advance for any one patient. Evaluating a patient’s response to the medication by spirometry is the only way to identify in advance those patients with COPD who will be helped by adding inhaled steroids to their regular regimen of bronchodilators (GOLD, 2025; Harding et al., 2022).
ANTI-INFLAMMATORIES
Corticosteroids
- Action: disrupts inflammatory pathways
- Drugs:
- Inhaled: fluticasone (Flovent)
- Oral: prednisone (Deltasone)
- Side effects:
- Inhaled: coughing, hoarseness, dry mouth, sore throat
- Oral: glaucoma, edema, hypertension, mood swings, weight gain, cataracts, hyperglycemia, infections, osteoporosis and fractures, menstrual irregularities, suppressed adrenal gland hormone production, thin skin, easy bruising, slower wound healing
- Comments: must be taken every day, even if there are no symptoms; not to be stopped suddenly; taken with food; reduces local immunity and may increase risk for local infections like candida (yeast)
Nonsteroidal anti-inflammatory drugs (NSAIDs)
- Action: stabilizes mast cell membranes to prevent inflammation
- Drugs: nedocrimil (Tiladel)
- Side effects: dyspepsia, nausea, hyperacidity; in higher doses, MI, CVA, rash, gastrointestinal bleeding
- Comments: must be taken by inhaler every day for prophylaxis, even if there are no symptoms; further testing needed to support the use of nedocrimil in COPD
(GOLD, 2025; Harding et al., 2022)
PREMIXED COMBINATION INHALERS
- Action: combines the effects of bronchodilators, corticosteroids, and anticholinergics
- Drugs:
- Short-acting: ipratropium and albuterol (DuoNeb), ipratropium and fenoterol (DuoVent)
- Long-acting: formoterol and budesonide (Symbicort); salmeterol and fluticasone (Advair); fluticasone furoate, umeclidinium bromide, and vilanterol (Trelegy Ellipta)
- Side effects:
- Short-acting: cough, dry mouth, tachycardia, palpitations, anxiety, muscle tremors
- Long-acting: coughing, hoarseness, dry mouth, sore throat, tachycardia, palpitations, anxiety, muscle tremors, headache, back pain, diarrhea, and altered taste
(GOLD, 2025; Harding et al., 2022; Medscape, 2024)
MUCOLYTIC AGENTS
Patients with COPD often have thick, tenacious mucus that is difficult to expectorate, particularly during an acute exacerbation. Mucolytic agents can be given by respiratory nebulizer treatments, sometimes mixed with normal saline to thin secretions. They can also be given orally to produce a systemic effect. Acetylcysteine (Mucomyst) or dornase alfa (Pulmozyme) are commonly given by inhaled nebulizer treatment, and guaifenesin (Robitussin) is given by mouth to promote expectoration.
MUCOLYTIC AGENTS
- Action: thins secretions to promote expectoration
- Drugs:
- Inhaled: acetylcysteine (Mucomyst), dornase alfa (Pulmozyme)
- Oral: guaifenesin (Robitussin)
- Side effects: foul smell, sticky nebulizer mask, white patches or sores inside mouth or on lips, nausea, fever, nasal drainage, sore throat, drowsiness, rash, clammy skin
- Comments: patient must be instructed in home nebulizer use; may interact with some vitamins, minerals, and herbs; may produce moderate improvement in health status and exacerbation reduction
(GOLD, 2025)
COVID-19 AND COPD TREATMENT
Some of the treatments for the COPD patient with a COVID-19 infection may be different than those usually employed, but for the most part, patients should take their oral and inhaled medication as usually directed. Nebulizers are used with caution because of the dispersal of droplets from contaminated aerosol or the patient coughing. COPD patients have been involved in clinical trials treating COVID-19 patients with new antiviral medications (GOLD, 2025).
ANTIBIOTICS
Studies show that continuous dosing with antibiotics will sometimes reduce exacerbations of COPD for patients particularly prone to exacerbations:
- Azithromycin 250 mg/day per os or 500 mg 3×/week for 1 year (less efficacy for active smokers), or
- Erythromycin 500 mg 2x/week for 1 year
(GOLD, 2025)
VACCINATIONS TO PREVENT COMPLICATIONS
People with COPD are at higher risk for serious, even life-threatening complications that are preventable by vaccination. As protection against serious lower respiratory illnesses, people with COPD are advised to receive an influenza vaccination each year. During outbreaks of strains of flu not covered by the annual vaccination, people with COPD should receive prophylactic antiviral treatment such as amantadine (Symmetrel), rimantadine (Flumadine), oseltamivir (Tamiflu), or zanamivir (Relenza). Older adult patients with COPD have decreased risk for ischemic heart disease when they have been vaccinated for the flu for several years.
Pneumococcal vaccinations are also recommended for people with COPD before age 65. A second and even third dose is recommended for people 65 years and older who got their first dose when they were younger than 65 if it has been five or more years since the first dose.
People with certain medical conditions such as COPD are the most likely to benefit from COVID-19 vaccinations. The COVID-19 virus can easily cause severe, life-threatening damage to lungs that are already at risk for respiratory distress, respiratory infection, and severe dyspnea. There has been extensive and conclusive evidence that those who are fully vaccinated against the COVID-19 virus tend to have milder episodes of the disease, and survival rates are much higher.
(GOLD, 2025)
ANSWERING PATIENT QUESTIONS
Q: Should I get a flu shot if I have COPD?
A: Flu can cause serious problems in people with COPD, and flu shots can reduce your chances of getting the flu. You should get a flu shot every year. In addition, you should have a pneumococcal vaccination, usually every five years, as well as a COVID-19 vaccination.
CASE
Rae Ann Evans presents to the urgent care clinic with a fever of 102.5 °F, (tympanic) diaphoresis, severe dyspnea with a respiratory rate of 28, a heart rate of 122, blood pressure of 158/92, and an oxygen saturation of 89% on room air. She is moderately obese. Upon assessment, the nurse auscultates her lungs and finds diminished breath sounds in the bases and expiratory wheezes throughout all fields.
The patient is sitting on the examination table bent forward, audibly wheezing, and using accessory chest muscles to breathe. She displays equilateral expansion of her chest. She states she is coughing up more secretions than usual and that they are dark yellow and thicker than normal.
Ms. Evans has chronic bronchitis-type COPD. She takes acetylcysteine (Mucomyst) to thin secretions to make them easier to bring up and the antibiotic azithromycin (Zithromax) every day to prevent infections, in addition to her daily bronchodilator inhalers. She is diagnosed with community-acquired pneumonia on top of her chronic COPD and given a nebulizer treatment, a prescription for an additional antibiotic to treat her pneumonia, and another prescription for a bronchodilator to be administered by nebulizer.
The clinician demonstrates the nebulizer machine to Ms. Evans and her husband, as they will have one delivered to their home for self-administered treatments until her condition improves. The clinician also discusses the current medication regime and the new additions, then questions the patient for an understanding on taking her meds correctly.
Oxygen Therapy
Supplemental oxygen improves levels of blood oxygenation and reduces the rate at which patients need to breathe. No other medical treatment has proved as effective in improving survival rates of patients with COPD. For people with COPD, supplemental oxygen also slows the rate at which muscles fatigue. These effects make it easier for patients to breathe more deeply and to exercise for longer periods.
Oxygen therapy is expensive and involves special equipment. Therefore, when people with COPD can maintain a blood oxygenation level of PaO2 >55–60 mmHg (an oxygenation saturation of more than about 89%), this is considered adequate oxygenation (unless the patient is symptomatic) and supplemental oxygen therapy is not routinely prescribed.
Long-term oxygen therapy (>15 hours/day) may be needed for stable COPD patients with an oxygen saturation <88% on room air, with or without hypercapnia. Stable patients may also need long-term oxygen therapy with an oxygen saturation <88% with pulmonary hypertension, peripheral edema, or polycythemia (hematocrit >55%) (GOLD, 2025).
A COPD patient’s dyspnea and concurrently poor arterial blood gasses (ABGs) (O2 saturation <90%, PaCO2 >45 mmHg, or pH <7.3), particularly low oxygen saturation, may necessitate the use of noninvasive ventilation. This may be established in the facility or home settings. When there is an acute exacerbation of the respiratory status in COPD, continuous positive airway pressure (CPAP) or bilevel positive airway pressure (BiPAP) may be used. These devices deliver pressurized oxygen through masks in order to maximize the delivery of needed oxygen deep in the lungs. The timely use of these devices, particularly in the prehospital period, has proved to significantly reduce the need for intubation and ventilation and to decrease mortality (UCSF, 2024).
High-flow nasal cannula (HFNC) oxygen therapy is increasingly and effectively used in patients with an exacerbation of COPD. The high-flow oxygen works to alleviate significant hypoxia during an exacerbation (Beuvon, 2022).
CONTINUOUS OXYGEN
Eventually, supplemental oxygen will be necessary for patients with COPD. For some patients, oxygen is needed to participate in regular exercise programs. For others, oxygen is needed simply to conduct typical ADLs.
If they live long enough, all patients with COPD lose sufficient lung function to the point that they will be hypoxemic at rest even on an optimal regimen of regular bronchodilator treatments. For these people, continuous oxygen therapy will not prolong their lives and reduce hospitalizations but will relieve or prevent breathlessness while at rest or during moderate activity (GOLD, 2025).
Low-flow (2–3 L/min) oxygen inhaled through nasal cannulas is usually sufficient to raise a COPD patient’s blood PaO2 to 65–80 mmHg (oxygen saturation of 89%–94%). In addition to increasing survival rates by about 50%, this level of supplemental oxygen lowers the person’s hematocrit toward a normal range, makes sleep easier, and improves exercise tolerance.
Home oxygen therapy is also recommended for patients with COPD with heart failure, pulmonary hypertension, or erythrocytosis (i.e., a hematocrit >56%) even when their PaO2 is >55 mmHg. Some patients who maintain a higher level of arterial oxygen during the day drop to a PaO2 <55 mmHg when they sleep. For people whose hemoglobin desaturates at night, nocturnal oxygen therapy is helpful.
HOME OXYGEN DELIVERY SYSTEMS
Home oxygen can be provided via an oxygen concentrator, via a compressed oxygen cylinder, or as liquid oxygen. All of these methods can supply an oxygen concentration of 90% or more to the individual and enrich the local environment. Medicare will pay for required durable medical equipment (DME) and gaseous or liquid oxygen for 36 months, with an additional 24 month extension, if necessary. In many cases, Medicare will cover 80% of the cost for supplemental oxygen for patients with desaturation (oxygen saturation <94%) during sleep or physical activity (Medicare.gov, 2024).
Patients usually breathe supplemental oxygen via a continuous-flow nasal cannula. Devices that “conserve” oxygen (reservoir cannulas such as moustache-configured oximizers or oximizer pendants, electromechanical demand pulse delivery devices, transtracheal oxygen delivery) are especially efficient because they provide all the supplemental oxygen early in each inhalation. Some patients who have trouble keeping low blood levels of carbon dioxide can be fitted with face masks from machines that deliver supplemental oxygen at continuous positive pressure; these systems provide noninvasive positive-pressure ventilation (Harding et al., 2022; Landry, 2025; Mondol et al., 2024).
A home system is usually adjusted to deliver oxygen at 2–3 L/min, and in most cases, this will maintain a patient’s oxygen saturation at >89%. For patients who continue to have dyspnea at night, the flow rate is raised by 1 L/min during sleep.
One goal of oxygen therapy is to allow patients to remain active. Inside the home, long tubes can connect the nasal cannulas to stationary oxygen delivery units so patients can move around. For more freedom and to go outdoors, patients can carry portable tanks of compressed oxygen or liquid oxygen. The risks of home oxygen therapy are hypercapnia, oxygen toxicity, and burns secondary to the flammable nature of oxygen (GOLD, 2025).
HAZARDS
Medical. There is a small risk that too high a concentration of inspired oxygen will suppress the respiratory drive (fueled by hypercapnia) of patients with COPD. Long-term low-flow oxygen therapy is probably safest when the amount of oxygen delivered gives the patient a PaO2 of 60–65 mmHg, which is toward the low end of the acceptable range of inspired oxygen.
Physical. Concentrated oxygen is flammable and poses a fire hazard. Patients and their families cannot smoke or use open flames near the oxygen equipment. The long oxygen tubing may also constitute a fall risk.
OXYGEN AND AIR TRAVEL
Commercial airplanes maintain an internal air pressure equivalent to 5,000–8,000 feet above sea level. For those patients with COPD whose resting arterial blood oxygen concentration is low (PaO2 <69 mmHg) even at sea level, the cabin concentration of oxygen will usually not be high enough to avoid hypoxemia. The PaO2 should be maintained at >50 mmHg by supplemental oxygen, if necessary, at 3 L/min per nasal cannula or at 31% by Venturi mask. Airlines can provide supplemental oxygen, and some airlines will allow patients to bring small oxygen delivery systems with them, although patients must make arrangements with the airline in advance (GOLD, 2025).
Surgery for COPD
Surgery is risky in people with severe COPD. Postoperatively, many normal patients temporarily have reduced lung volumes, rapid shallow breathing, and an impaired ability to take in oxygen and expel carbon dioxide. These routine postoperative problems add additional stress to the already compromised respiratory systems of patients with COPD. One result is that patients with severe COPD develop postoperative pneumonia 13 times more often than patients with normal lung function. Preoperative antibiotics can reduce the high rate of postoperative pneumonia.
The lack of alternative treatments for severe COPD has led to the development of three surgical procedures that attempt to improve and prolong the lives of patients with COPD. The techniques are lung transplantation, lung volume reduction surgery, and bullectomy.
LUNG TRANSPLANTATION
People with severe COPD are the most common recipients of lung transplants. Candidates for lung transplantation are patients with severe COPD who have exhausted medical therapies and have life expectancies of ≤2 years. The BODE Index is used to estimate a COPD patient’s life expectancy (see box below). Typically, patients should also be younger than 65 years. Three quarters of patients with COPD who receive lung transplants live for ≥2 years after the operation, and many of the survivors have substantially improved abilities to exercise.
BODE INDEX TO PREDICT SURVIVAL
The updated BODE Index uses four measurements (body mass index, airway obstruction, dyspnea, and exercise capacity) to assign patients with COPD to 1 of 15 groups, each with a different estimated survival rate.
This mortality prediction index is a multistage scoring system that provides prognostic data in patients with COPD. The BODE index is better than the FEV1 at predicting the risk of death among patients with COPD. The measurements are:
- B: body mass index
- >21 (0 points)
- ≤21 (1 point)
- O: degree of airflow obstruction (FEV1 calculated by spirometric measurement)
- ≥65% (0 points)
- 50%–64% (1 point)
- 36%–49% (2 points)
- ≤35% (3 points)
- D: amount of dyspnea (using the Modified Medical Research Council dyspnea scale)
- mMRC 0 = dyspneic on strenuous exercise (0 points)
- mMRC 1 = dyspneic on walking a slight hill (0 points)
- mMRC 2 = dyspneic walking level ground; must stop occasionally for breathlessness (1 point)
- mMRC 3 = must stop for breathlessness after walking 100 yards or a few minutes (2 points)
- mMRC 4 = cannot leave house; breathless on dressing/undressing (3 points)
- E: exercise capacity (distance walked in 6 minutes)
- ≥350 meters (0 points)
- 250–349 meters (1 point)
- 150–249 meters (2 points)
- ≤149 meters (3 points)
Four years after a BODE assessment is made, estimated survival rates for long-standing patients with COPD are approximately:
- 18% for a BODE score of 7–10
- 57% for a BODE score of 5–6
- 67% for a BODE score of 3–4
- 80% for a BODE score of 0–2
(Mosenifar, 2024)
LUNG VOLUME REDUCTION SURGERY
As noted earlier, the lungs of an emphysematous patient become hyperinflated with air spaces that contribute little to gas exchange. The widened chest caused by hyperinflated lungs is difficult for the patient to expand further when attempting to inhale. By removing lung tissue that contains dead air space, surgery can sometimes reduce the patient’s work of breathing by improving airflow obstruction.
In lung volume reduction surgery, the damaged lung tissue is removed from both sides of the chest. As a result, survivors can usually exercise more than they could before the surgery. Those patients who have mainly upper-lung emphysema also have an increased lifespan after this surgery. Exclusion criteria include ≥75 years of age, active smoker, or unable to exercise despite pulmonary rehabilitation (PR).
The major postoperative complication of lung volume reduction surgery is continuing air leakage from the lungs into the chest. Other potential complications are pneumothorax, pneumonia, blood clots, infection, myocardial infarction, dysrhythmias, or the formation of a fistula (ALA, 2024e).
BULLECTOMY
In some cases, individual large empty air spaces (bullae) can be surgically removed. Typical bullae in a patient with emphysema are a few centimeters in diameter. Occasionally, however, bullae can be huge, taking up as much as a third of the chest space. These giant bullae squeeze the healthier lung tissue and compress the adjacent blood vessels. By removing giant bullae, the remaining lung tissue can reexpand, and some of the circulation will be restored.
As with lung volume reduction surgery, a major postsurgical complication of bullectomy is persistent air leakage. Premedication with antibiotics greatly reduces the postoperative incidence of pneumonia (Harding et al., 2022).
ZEPHYR VALVE
A Zephyr valve is a one-way valve designed to improve breathing and reduce shortness of breath in a patient with COPD by inducing an artificial atelectasis in the most diseased part of the lung. It is placed in the airway by bronchoscopy in a minimally invasive, nonsurgical procedure. The valve promotes the expiration of air, allowing the lung to empty, but prevents more air from entering the lung and causing it to hyperinflate. This allows the healthier areas of the lung to better function. The most common side effect of this procedure is a pneumothorax. Other possibilities are exacerbation of COPD symptoms, pneumonia, and hemoptysis (Pulmonx, 2025).
ACUTE EXACERBATION OF COPD
Patients with COPD have little or no ventilatory reserve, and a further compromise of their respiratory system can send them into hypoxemia. The normal wear and tear of daily life puts respiratory compromises in everyone’s path periodically. People with COPD respond poorly to these respiratory problems and often experience an increase in dyspnea, cough, and sputum production. Such episodes of suddenly worsening symptoms are called acute exacerbations. Acute exacerbations usually last seven to 10 days, but 20% of COPD patients may still be experiencing increased symptoms for up to eight weeks.
The significance to healthcare personnel of patients’ exacerbations is their deleterious effect on patients’ health, the healthcare economy, increased hospital readmissions, accelerated rate of disease progression, future exacerbation risk, and mortality (GOLD, 2025; Harding et al., 2022).
Causes of Acute Exacerbations
Acute exacerbations of COPD can be brought on by a variety of factors. Infections, especially respiratory infections from colds to pneumonias, are common triggers. Smoking, passive smoking, reactivity of airways, occupational factors, and air pollution are the risk factors of COPD and can be the cause of an acute exacerbation.
Acute exacerbations can also be triggered by other medical conditions, especially when these conditions impinge on the cardiovascular or respiratory systems. Pneumothorax, pulmonary emboli, congestive heart failure, heart arrhythmias, chest trauma, lung atelectasis, and pleural effusions will all worsen a patient’s COPD. At the same time, however, many acute exacerbations cannot be easily explained.
Signs and Symptoms of an Acute Exacerbation
During an acute exacerbation, patients become more breathless than usual. They may have chest tightness, begin to wheeze or cough, and find it difficult to talk. In addition, their airways can become clogged with sputum, which may be yellowish or greenish and filled with white blood cells.
A sudden decrease in the ability to breathe efficiently makes patients tachycardic and sweaty, and their percentage of oxygenated hemoglobin (measured by pulse oximetry) decreases. In serious cases, patients become hypercapnic because they cannot get rid of sufficient carbon dioxide, making them acidotic and lethargic.
Treatment of an Acute Exacerbation
A patient’s regularly scheduled medications will not reverse an acute exacerbation. Instead, extra rescue medicines (a short-acting bronchodilator) and systemic corticosteroids are needed. To prevent ventilatory decompensation from worsening, further medical assistance, including hospitalization and mechanical ventilation, can be needed to treat an acute exacerbation and its cause (Harding et al., 2022).
Unlike asthma exacerbations, which can usually be reversed quickly, acute exacerbations of COPD improve slowly even when the patient receives prompt medical care. On average, it will take a week for a person to recover from an exacerbation of COPD, and recovery from 1 out of 4 acute exacerbations takes more than a month. For patients with severe COPD, an acute exacerbation can be fatal.
RESCUE MEDICATIONS
As a first step in counteracting the sudden worsening of their lung functions, patients are usually advised to take a predetermined “rescue dose” of a short-acting bronchodilator. Typically, it is a beta-2 agonist (albuterol, pirbuterol, or terbutaline), ipratropium, or the combination of albuterol and ipratropium (GOLD, 2025; Harding et al., 2022). Patients with COPD are advised to always keep their quick-relief inhaler with them.
EMERGENCY EVALUATION
When a sudden worsening of the ability to breathe is not improved by rescue therapy, the patient must be seen quickly by a primary care provider. Besides their COPD, the patient could be experiencing a medical emergency such as pneumothorax, pulmonary embolism, anaphylaxis, airway obstruction, or myocardial infarction. When the person with COPD does not improve with the usual rescue medications or home oxygen (if available), the patient or family should call the physician or 911 or report to the emergency department, depending on the severity of symptoms.
Anyone with the sudden onset of severe dyspnea is evaluated as a medical emergency. First, it must be ascertained that the patient has a clear airway. Diagnostic tests may include chest X-ray, electrocardiography, arterial blood gasses, complete blood count, electrolytes, cardiac enzymes, peak flow measurement, pulse oximetry, and pulmonary function testing (Harding et al., 2022).
At the same time, an intravenous line is established and a cardiac monitor connected. If the patient’s pulse oximetry shows an oxygen saturation of <88%, supplemental oxygen is given, with a target oxygen saturation of 88%–92% (GOLD, 2025; Harding et al., 2022). The possibility of a pulmonary embolus is always considered when there is a sudden increase in dyspnea and hypoxia.
The patient should be medically stabilized. Patients with a serious instability or decompensation are admitted to an intensive care unit and the workup continues there. In this case, there is serious consideration of ventilatory support. This is a decision made in conjunction with the patient, if possible, or the patient’s family, since there is a strong possibility that the patient may not be able to be weaned from the ventilator and extubated.
MEDICAL MANAGEMENT
For patients experiencing an acute exacerbation of COPD, the immediate goals are to maintain an adequate level of blood oxygen and an appropriate blood pH.
For some patients with COPD, their exacerbation will be sufficiently mild that bronchodilators, steroids, and oxygen will lead to a rapid improvement. Other pharmacologic treatments may include a short-acting beta agonist, a short-acting inhaled anticholinergic, corticosteroids, and antibiotics. If no treatable trigger is found for this episode, the patients can often be sent home and followed outside the hospital (Harding et al., 2022).
Other patients’ lung functioning will have deteriorated sufficiently so that these persons must be supported in a hospital. COPD leads to chronic respiratory failure, and acute exacerbations can lead to the superposition of acute respiratory failure. The result is called acute-on-chronic respiratory failure. In acute-on-chronic respiratory failure, patients have increasing dyspnea and may eventually develop an altered mental state or even respiratory arrest. Acute-on-chronic respiratory failure typically produces an acidosis, with pH <7.35 (normal pH is 7.35–7.45).
For acute-on-chronic respiratory failure patients, hospital therapy includes bronchodilator treatments, systemic steroids, controlled oxygen, and often intravenous antibiotics. When necessary, steps are taken to maintain the patient’s ventilation and circulation. Supplemental oxygen for hypoxemia in patients with COPD is given to keep blood oxygenation levels of 88%–92%. Meanwhile, attempts are made to identify and reverse the precipitating factors; if a specific infection has not been identified, antibiotics are sometimes given prophylactically (GOLD, 2025; Harding et al., 2022).
In severe cases, noninvasive positive pressure mechanical ventilation (also called noninvasive ventilatory support, or NIVS) with a face mask or nasal cannula will often improve gas exchange without having to intubate the patient. Noninvasive ventilation leads to fewer secondary pneumonias and is easier to wean patients from than endotracheal intubation (GOLD, 2025).
ANTIBIOTICS FOR ACUTE EXACERBATIONS OF COPD
Respiratory infections are frequent causes of acute exacerbations of COPD. Likely microbes include Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis, and Pseudomonas aeruginosa.
When an acute exacerbation includes signs of infection (e.g., fever, elevated white blood cell count, purulent sputum, or a suggestive chest X-ray), the empirical administration of antibiotics is usually recommended. The most commonly ordered classifications of antibiotics are aminopenicillin with clavulanic acid, macrolides, and tetracycline.
Antibiotics reduce the risk of short-term mortality by 77%, treatment failure by 53%, and sputum purulence by 44%. Inflammation caused by an exacerbation of COPD increases the risk of respiratory infection. The lab test C-reactive protein (CRP) measures the level of inflammation in general. One study showed that when the CRP is low, the use of antibiotics is reduced from 77.4% to 47.7% (GOLD, 2025).
ANSWERING PATIENT QUESTIONS
Q: How do I know when to call for emergency help for my COPD?
A: People with COPD will have episodes called acute exacerbations. During these episodes, you will have a much harder time catching your breath. You may also experience chest tightness, more coughing, a change in your sputum, or a fever. It is important to call your primary care provider if you have any of those signs or symptoms. Specifically, you should get emergency help or advice if:
- You have taken your rescue medicines and you still feel as if you can’t breathe
- You find that it is suddenly hard to talk or to walk
- You are coughing up more mucus and it is yellow, green, or brown
- You develop a fever
- You get unusual chest pain or chest tightness
- Your heart is beating very quickly or irregularly for more than a few minutes
- Your lips or fingernails are gray or blue
- Your breathing is fast and hard, even after you have used your medicines
- Your mind is getting cloudy or you are getting tired and sleepy at the wrong time
Because it is likely that you will have an acute exacerbation at some time, be prepared. Plan now and have these things easily available:
- Your rescue medicines for sudden spells of difficult breathing
- Phone numbers of your primary care provider and of people who can take you to your primary caregiver’s office or to a nearby emergency department
- Directions to your primary caregiver’s office and to a nearby emergency department
- A list of the medicines that you usually take
Q: What should I do if I am having more trouble than usual catching my breath or if I am coughing more than usual?
A: If you have a set of rescue medicines that you have been told to take, go ahead and use them. Then call your primary care provider right away.
Q: What do I do when I’m getting sick, like with a fever or a cold?
A: Call your primary care provider right away.
END-STAGE CARE
It is important for patients with COPD, families, and primary care providers to discuss end-of-life issues. Advance directives, living wills, and durable powers of attorney can be explained and decided upon. Many states now require that hospitalized patients be given the opportunity to receive information about advance directives in particular.
It is often difficult for patients, family members, or close friends to contemplate these serious decisions that need to be made for someone with a diagnosis that is uncurable and eventually terminal. The subject of invasive and noninvasive ventilation is a particularly necessary subject for this patient population. The multidisciplinary team can be honest, matter-of-fact, and sensitive in conversations about what choices need to be made. Chaplains and personal clergy can be brought in for additional support.
Decisions that patients and their families will face include whether to participate in drug trials, what type of ventilation to use and for how long, whether to consider lung transplantation, whether to take advantage of hospice, and what type of end-of-life palliation is desired. Counseling may be provided by specially trained hospice nurses or in-hospital palliative care nurses.
Palliative care becomes a valuable adjunct of medical care in patients with COPD in the absence of curative treatment. This means that maintaining and, when possible, improving a patient’s quality of life should always be a prime motivator of therapy. Patients with COPD experience psychological and emotional suffering and high morbidity and mortality rates. In spite of this, they do not always receive adequate palliative care.
Palliative care is a rapidly growing, multidisciplinary specialty that is used to improve quality of life for seriously ill patients and their families. The various disciplines involved include physicians, nurses, social workers, chaplains, and other specialties, when appropriate. Early palliative care also means that patients and their families should be encouraged to consider end-of-life options early in the disease process, before the patient becomes mentally compromised or the family becomes emotionally worn out (Fusi-Schmidhauser, 2021).
Too often COPD patients die in intensive care units after invasive, extensive, expensive, and often unwanted medical care. Near the end of life, adoption of hospice care will give the patient and their family the opportunity to discuss available options and the extent to which the patient wishes to be treated at the end of their life. This discussion may lead to the decision to receive palliative care at home, which can reduce unnecessary hospitalizations and allow the patient to die in comfortable and familiar surroundings.
CONCLUSION
COPD is one of the leading causes of death in the United States and worldwide, caused primarily by cigarette smoking. It leads to breathing difficulty even with mild exercise and during routine activities of daily living, eventually making it hard for the person with COPD to do anything more than sit or lie down.
Healthcare professionals can encourage and support all patients to stop smoking immediately as a form of prevention or, in those with COPD, to slow down the progression of the disease and improve their quality of life. COPD’s first symptom is typically coughing, although dyspnea is what usually first prompts someone to seek care.
COPD is a lifelong disease, and treatment focuses both on long-term goals (slowing its progression, easing symptoms, improving quality of life, preventing acute exacerbations) and managing acute exacerbations. Therapeutic lifestyle changes are fundamental, and healthcare professionals can assist patients though education, supporting smoking cessation, and pulmonary rehabilitation programs. There is no cure, but pulmonary rehabilitation has been effective in improving COPD patients’ physical function and endurance. Patients may, at best, prolong the gradual decline in their ability to perform activities of daily living over the course of their remaining lifetime.
Primary care providers introduce drug therapy to reduce restrictions in a patient’s activities. Bronchodilators are used to relieve dyspnea and are typically administered via inhalers. Corticosteroids are also effective as anti-inflammatories, generally used in patients with severe COPD. Eventually, oxygen therapy may be required. Surgery (lung transplantation, lung volume reduction, and bullectomy) may be also recommended in cases of severe COPD that no longer respond to medication therapies.
There are special considerations for the COPD patient who is COVID-19 positive. While COPD patients are not found to be at greater risk for contracting the SARS-CoV-2 virus, once infected they are at greater risk for exacerbation of their chronic illness because their lungs are more fragile. It is therefore especially important that COPD patients follow COVID-19 prevention precautions. Some COPD treatments must also be altered in patients with COVID.
Because COPD is a chronic condition affecting millions of people, healthcare providers can provide effective care by helping their patients understand the disease, quit smoking in order to slow its progression, learn how to manage their long-term treatment, and prepare for and know what actions to take in the event of an acute exacerbation.
RESOURCES
Chronic obstructive pulmonary disease (COPD) (American Lung Association)
COPD clinical resources (American Thoracic Society)
Freedom from smoking (American Lung Association)
GOLD (Global Initiative for Chronic Obstructive Lung Disease)
Health effects of cigarettes: COPD (Centers for Disease Control and Prevention)
What is COPD? (National Heart, Lung, and Blood Institute)
REFERENCES
NOTE: Complete URLs for references retrieved from online sources are provided in the PDF of this course.
American Lung Association (ALA). (2024a). COPD trends brief: Prevalence. https://www.lung.org
American Lung Association (ALA). (2024b). E-cigarette or vaping use–associated lung injury. https://www.lung.org
American Lung Association (ALA). (2024c). The impact of e-cigarettes on the lungs. https://www.lung.org
American Lung Association (ALA). (2024d). Conserving energy and managing your daily activities. https://www.lung.org
American Lung Association (ALA). (2024e). Lung volume reduction surgery. https://www.lung.org
American Lung Association (ALA). (2023a). Physical activity and COPD. https://www.lung.org
American Lung Association (ALA). (2023b). COPD screening, assessment, and prediction tools. https://www.lung.org
Awell. (2024). Mini-best test. https://score.awellhealth.com
Beuvon C, Coudroy R, Bardin J, Marjanovic N, Rault C, Bironneau V, Drouot X, Robert R, Thille AW, & Frat JP. (2022). β agonist delivery by high-flow nasal cannula during COPD exacerbation. Respiratory Care, 67(1), 9–15. https://doi.org/10.4187/respcare.09242
Canadian Association of Occupational Therapists (CAOT). (2024). About the COPM. https://www.thecopm.ca
Centers for Disease Control and Prevention (CDC). (2024a). Youth and tobacco use. https://www.cdc.gov
Centers for Disease Control and Prevention (CDC). (2024b). About e-cigarettes. https://www.cdc.gov
Centers for Disease Control and Prevention (CDC). (2024c). E-cigarette use among youth. https://www.cdc.gov
Centers for Disease Control and Prevention (CDC). (2024d). Health effects of cigarettes: Chronic obstructive pulmonary disease (COPD). https://www.cdc.gov
Centers for Disease Control and Prevention (CDC). (2024e). Leading causes of death. https://www.cdc.gov
CESD-R. (2024). Center for epidemiologic studies depression scale revised. http://cesd-r.com
Cleveland Clinic. (2024). Berg balance scale. https://my.clevelandclinic.org
COPD Foundation. (2024). Breathing techniques. https://www.copdfoundation.org
Fusi-Schmidhauser T. (2021). Palliative care for patients with advanced COPD in primary care. Praxis, 110(15), 902–6. https://doi.org/10.1024/1661-8157/a003790
GeneCard. (2024). ACTA2 gene—actin alpha 2, smooth muscle. https://www.genecards.org
Global Initiative for Chronic Obstructive Lung Disease (GOLD). (2025). Global strategy for the diagnosis, management and prevention of chronic obstructive pulmonary disease: 2025 report. https://goldcopd.org
Haddad M & Sharma S. (2023). Physiology, lung. https://www.ncbi.nlm.nih.gov
Harding MM, Kwong J, Roberts D, Hagler D, & Reinisch C. (2022). Lewis’s medical-surgical nursing: Assessment and management of clinal problems (12th ed.). Mosby.
Healthline. (2024). COPD drugs: A list of medications to help relieve your symptoms. https://www.healthline.com
Koolen EH, Spruit MA, de Man M, Antons JC, Nijhuis E, Nakken N, et al. (2021). Effectiveness of home-based occupational therapy on COPM performance and satisfaction scores in patients with COPD. Canadian Journal of Occupational Therapy, 88(1), 26–37.
Landry J. (2025). Oxymizer: Uses, types, benefits, and disadvantages. Respiratory Therapy Zone. https://www.respiratorytherapyzone.com
LoMauro A & Gervasoni F. (2024). 20 years of neuromuscular electrical stimulation in COPD. European Respiratory Review, 33(171), 220247. https://doi.org/10.1183/16000617.0247-2022
McCuistion LE, DiMaggio KV, Winton MB, & Yeager JJ. (2022). Pharmacology: A patient-centered nursing process approach (11th ed.). Saunders.
McMillen M. (2023a). What are pink puffers and blue bloaters in COPD? HealthCentral. https://www.healthcentral.com
McMillen M. (2023b). How a tripod position can help with COPD. HealthCentral. https://www.healthcentral.com
MDCALC. (2024). Barthel index for activities for daily living (ADLs). https://www.mdcalc.com
Medicare.gov. (2024). Oxygen equipment and accessories. https://www.medicare.gov
Medscape. (2024). Fluticasone furoate/umeclidinium bromide/vilanterol inhaled (Rx). https://reference.medscape.com
Mosenifar Z. (2024). What is the BODE index, and how is used to estimate the prognosis of chronic obstructive pulmonary disease (COPD)? Medscape. https://www.medscape.com
Mondal A, Dutta D, Chanda N, Mandal N, & Mandal S. (2024). RESPIPulse: Machine learning assisted sensory device for pulsed mode delivery of oxygen bolus using surface electromyography (sEMG) signals. Sensors and Actuators A: Physical, 369, 115121. https://www.sciencedirect.com
National Institutes of Health (NIH). (2024a). What is COPD? https://www.nhlbi.nih.gov
National Institutes of Health (NIH). (2024b). COPD national action plan at-a-glance. https://www.nhlbi.nih.gov
National Institutes of Health (NIH). (2022). COPD: A national health issue fact sheet. https://www.nhlbi.nih.gov
Pagana K, Pagana T, & Pagana T. (2022). Mosby’s manual of diagnostic and laboratory tests (7th ed.). Elsevier.
Pulmonx. (2025). Learn how the Zephyr valve may help you. https://uspatients.pulmonx.com
Richards JB. (2024). mMRC (Modified Medical Research Council) dyspnea scale. https://www.mdcalc.com
Sandhu A, Hosseini SA, & Saadabadi A. (2023). Nicotine replacement therapy. StatPearls Publishing. https://www.ncbi.nlm.nih.gov
Su WC, Juan HL, Lee JI, Huang SP, Chen SC, & Geng JH. (2024). Secondhand smoke increases the risk of developing chronic obstructive pulmonary disease. Scientific Reports, 14, 7481. https://doi.org/10.1038/s41598-024-58038-2
Tee-Melegrito RA. (2023). What are the odds of surviving COVID-19 with COPD? https://www.medicalnewstoday.com
Tomashaw B & Carter C . (2023). Closing the gap: Identifying and treating COPD in women. COPD Foundation. https://www.copdfoundation.org
Tounsi B, Acheche A, Lelard T, Tabka Z, Trabelsi Y, & Ahmaidi S. (2021). Effects of specific inspiratory muscle training combined with whole-body endurance training program on balance in COPD patients: Randomized controlled trial. PloS one, 16(9), e0257595. https://doi.org/10.1371/journal.pone.0257595
United Kingdom Research and Innovation (UKRI). (2022). MRC dyspnoea scale. https://www.ukri.org
University of California, San Francisco (UCSF). (2024). Noninvasive positive pressure ventilation (NIPPV) BiPAP or CPAP. https://hospitalhandbook.ucsf.edu
World Health Organization (WHO). (2023). Tobacco. https://www.who.int/news-room/fact-sheets/detail/tobacco
Yamaguchi T, Yamamoto A, Oki Y, Sakai H, Misu S, Iwata Y, et al. (2021). Reliability and validity of the Japanese version of the Barthel Index dyspnea among patients with respiratory diseases. International Journal of Chronic Obstructive Pulmonary Disease, 16, 1863.
Zhang L, Liu Y, Zhao S, Wang Z, Zhang M, Zhang W, Hao L, & Jiao G. (2022). The incidence and prevalence of pulmonary hypertension in the COPD population: A systematic review and meta-analysis. International Journal of Chronic Obstructive Pulmonary Disease, 10(17), 1365–79. https://doi.org/10.2147/COPD.S359873






Customer Rating
4.9 / 548 ratings